- Home
- Adherent HEK293t Cells Cultured in Pall’s iCELLis® Bioreactor with OptiPEAK HEK293t Blood-Free Chemically Defined Media Exhibit Robust and Rapid Population Doubling Times
Adherent HEK293t Cells Cultured in Pall’s iCELLis® Bioreactor with OptiPEAK HEK293t Blood-Free Chemically Defined Media Exhibit Robust and Rapid Population Doubling Times
Published on 1 September 2020
Andrew Laskowski1 , Randy Alfano2 , Annie Cunningham2 , Atherly Pennybaker2 , Todd Lundeen1 1 Pall Biotech, 20 Walkup Drive, Westborough, Massachusetts 01581, USA ; 2 Invitria, 12635 E Montview Blvd, Aurora, Colorado 80045, USA
ICELLIS BIOREACTOR BACKGROUND
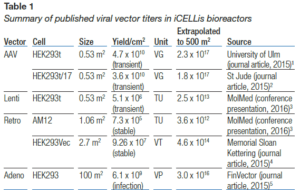
In the past five years, Pall’s iCELLis bioreactor has emerged as the leading bioreactor technology for clinical and commercial manufacturing of viral vectors and vaccines, with the following data reported in peer- reviewed publications.
- Single-use, fixed-bed bioreactor
- Cell substrate composed of medical grade, uncoated, uncharged polyethylene teraphalate (PET) carriers
- Closed system with reduced footprint and minimal aseptic handling
- Bench and commercial-size scalability by maintaining fixed-bed height and carrier compaction
- pH and dissolved oxygen (DO) control through a falling film
- High gas transfer rates due to large surface area and thin film mass transfer, no sparging required
- No shear due to rising bubbles or bubble burst
- Less base addition required due to high CO2 stripping capacity
- Compartmentalization – base addition, agitation, and fixed-bed spatially separate
- Reduced shear stress from impellor energy dissipation
- Evenly distributed media circulation
- No fixed-bed pH heterogeneity
- Fixed-bed acts as natural cell retention device for perfusion

INVITRIA OPTIPEAK293T BACKGROUND
- Chemically defined media that is optimized specifically for the adherent HEK293t-based viral vector production in the iCELLis bioreactor.
- Developed to circumvent the high variability and cost found in classical serum supplemented media for virus production.
- Utilizes a recombinant human transferrin and albumin from a large scale non mammalian expression platform and therefore does not
- incorporate any human or blood-derived components.
METHODS
- Pre-adapted HEK293t cells were expanded for 3 passages with each passage being seeded at 10,000 cells/cm2 and flasks subcultured for 3-4 days.
- iCELLis Nano bioreactor settings for OptiPEAK293t were optimized based on growth characteristics from previous studies (Table 2).
- Future experiments will include equivalent settings for both SFM and FBS containing media.
- Cell inoculations consisted of 100 mL of growth medium containing
- 5.3 x 107 cells followed by an additional 100 mL of growth medium in order to completely flush the inoculation line. Time of inoculation is considered t = 0.
- During the course of the runs, bioreactors were subjected to two 100% media changes, one at Day 3 and the transfection at Day 7.
- Data is presented as percent of the original (for the attachment kinetics only) 5.3 x 107 cell inoculum still present in the media vessel of the bioreactor at the time of sample pull.
- On day 7, cells were transfected with phMGFP vector complexed with polyethylenimine (0.15 μg/cm2 DNA at a 1:5 ratio with PEI).
- Vector was complexed in basal DMEM/F12 for 30 minutes at room temperature and then added to fresh growth medium. Transfections were done by circulating 600 mL of the DNA/PEI complex in growth medium, through the fixed-bed, for 1 hour.
- Media volume was brought up to 903 mL with fresh growth medium. Bioreactors were run for an additional 48 hours post transfection with standard growth parameters being monitored.
RESULTS
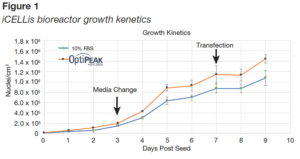
- Doubling times were lower in OptiPEAK293t, (Figure 1 and Table 3), resulting in higher cell density on the day of transfection. We hypothesize this better cell growth is due to cells acclimating to the fixed-bed faster in OptiPEAK293t.
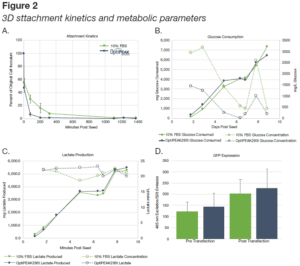
- Cells attached more rapidly to the carriers in OptiPEAK293t than in DMEM+10% FBS.
- By ~18 hours (1111 minutes) post inoculation (Figure 4A), >99% of all cells had attached in both bioreactors.
- Data is presented as percent of the original 5.3 x 107 cell inoculum still present in the media vessel of the bioreactor at the time of sample pull (Figure 4A).
- Glucose consumption and lactate production increased as biomass increased in the bioreactors (Figure 2B-C). Cells in OptiPEAK293t exhibited greater glucose consumption than DMEM+10% FBS for the first 4 days and subsequently the same glucose consumption as FBS containing media.

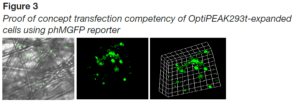
- A significant increase in the expression of phMGFP was detected in samples removed post-transfection (Figure 2D). To confirm these findings, fluorescent microscopy was employed to determine the presence of phMGFP-positive cells in the iCELLis bioreactor carriers harvested. phMGFP+ cells were found in multiple layers of the carrier with scanning sections 85 μm thick (Figure 3).
CONCLUSION
- These studies suggest that chemically defined, serum free media such as OptiPEAK293t is compatible with the iCELLis bioreactor platform and is a viable alternative to the use of FBS-containing media.
- HEK293t cells exhibited doubling times that were commercially viable and comparable to FBS containing media. OptiPEAK293t induced rapid and irreversible cell attachment to the iCELLis bioreactor carriers and were readily transfected by the reporter vector in a simplified transfection protocol.
- Multiple layers of cells within the iCELLis bioreactor carriers harvested from iCELLis Nano bioreactor were positive for the reporter, indicating a high degree of penetration of vector throughout the carriers in the OptiPEAK293t containing media.
The following content is gated. Please, subscribe to open access to it.