- Home
- Beyond Synthetic Surfactants: The Case for Recombinant Human Serum Albumin (rHSA) as an Ideal Alternative to Poloxamer 188
Beyond Synthetic Surfactants: The Case for Recombinant Human Serum Albumin (rHSA) as an Ideal Alternative to Poloxamer 188
Published on 25 March 2026
Article
Marcus Curl, VP of Product Applications
Reading time: 4 minutes
Share this article:
The Poloxamer 188 Crisis: Synthetic Variability and Risk
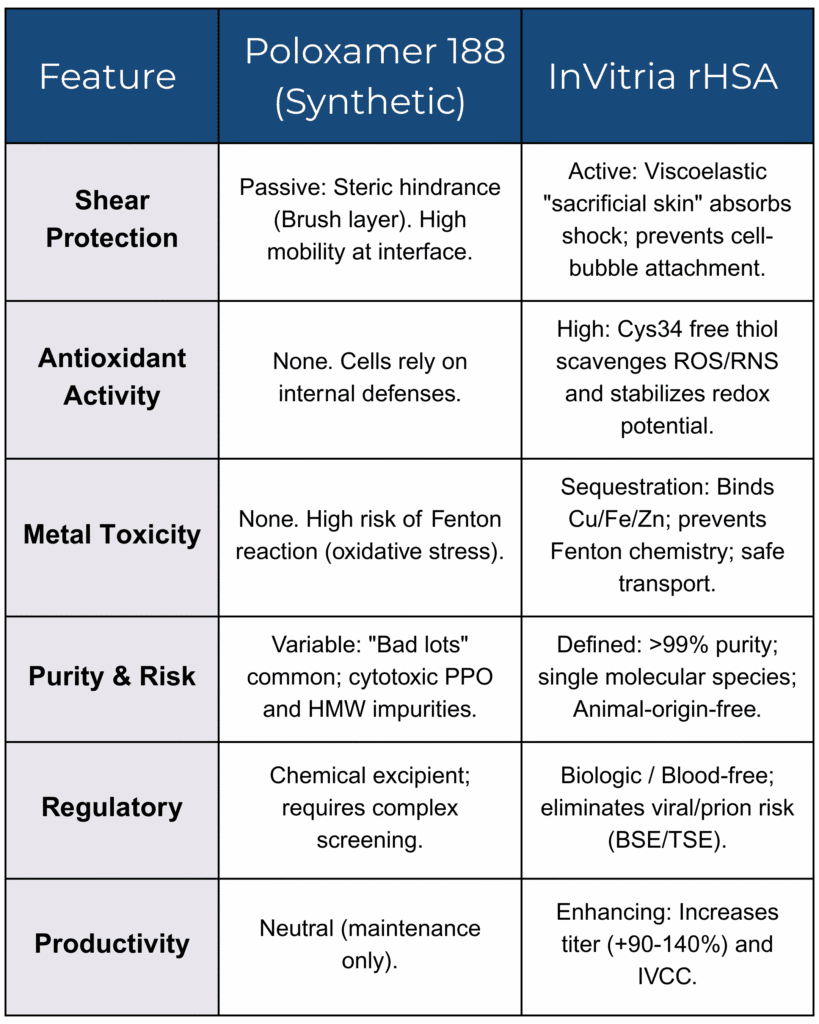
The inclusion of Poloxamer 188 challenges the definition of “chemically defined” media. Although often described as a triblock copolymer (PEO-PPO-PEO), commercial P188 is a statistical distribution of polymer chains, rather than a single, defined molecule. This heterogeneity has direct consequences for process robustness.
Lot-to-Lot Variability and Manufacturing Failure
Confidence in P188 was shattered by multiple investigations into “bad lots” associated with severe reductions in CHO cell viability and productivity.
- The Biogen investigation: In a landmark series of studies, researchers identified that specific lots of P188 that met all standard compendial USP/NF specifications, nevertheless failed in cell culture. The root cause was identified as trace hydrophobic impurities, specifically high-molecular-weight (HMW) species. These impurities preferentially adsorbed to gas-liquid interfaces but failed to provide effective steric protection (Peng et al., 2014; Peng et al., 2016).
- Foam stability as a marker: Problematic P188 lots were characterized by unusually stable foam. This behavior indicated the presence of rigid layers formed by HMW impurities that trapped cells rather than repelling them, increasing shear-induced cell damage (Peng et al., 2016).

Cytotoxicity of Polypropylene Oxide (PPO)
Further analysis has revealed that incomplete polymerization during manufacturing can leave residual polypropylene oxide (PPO) homopolymer in P188 formulations. Unlike the amphiphilic triblock copolymer, PPO is highly lipophilic. It partitions into CHO cell membranes, disrupts lipid raft architecture, and alters membrane fluidity. These effects lead to cytostasis and apoptosis, even in the absence of shear stress (Bandyopadhyay et al., 2022).
As a result, manufacturers are forced to rely on expensive biological use-tests to screen P188 lots, adding risk, expense and operational burden to the supply chain.
Mechanism of Action: The Biological Superiority of rHSA
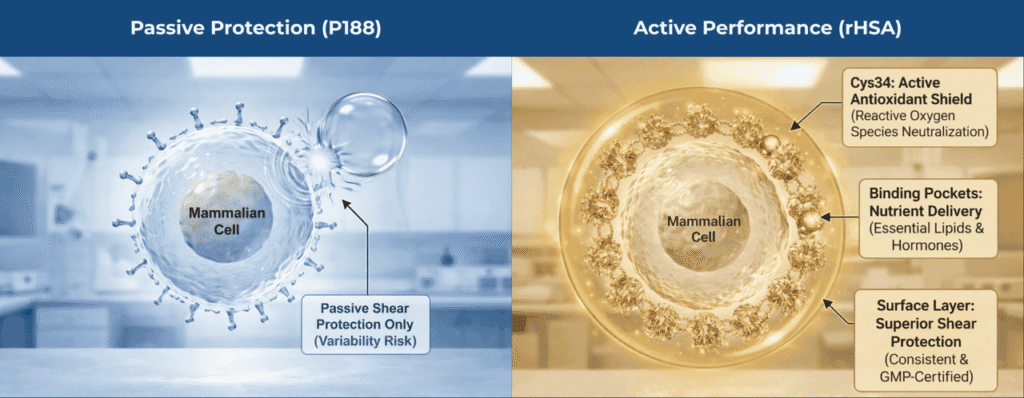
Poloxamer 188 provides one single function: reduction of surface tension through steric hindrance, a “brush” layer. Albumin, by contrast, evolved to preserve cellular homeostasis under turbulent vascular flow and offers multiple protective mechanisms that P188 cannot replicate.
Viscoelastic “Sacrificial Skin” vs. Steric Hindrance
The human immune system is evolutionarily tuned to recognize particulate matter as a “danger signal.”
- P188 mechanism: P188 surfactants are highly mobile. When a bubble bursts, they rapidly clear from the interface through Marangoni-driven flow, offering minimal mechanical resistance to the resulting shock wave.
- Albumin mechanism: Albumin forms a viscoelastic protein layer at the air-liquid interface. This “sacrificial skin” absorbs and dissipates the kinetic energy during bubble rupture, reducing hydrodynamic stress before it reaches the cell (Garcia-Briones & Chalmers, 1992). Furthermore, albumin coats the cells themselves, masking hydrophobic membrane patches and preventing cell-to-bubble attachment, effectively keeping cells out of the “kill zone” (Francis, 2010).
Antioxidant Defense (The Cys34 Thiol)
High-density CHO cultures generate substantial reactive oxygen species (ROS). P188 is chemically inert and offers no antioxidant protection.
- Cys34 scavenging: Albumin is the primary extracellular antioxidant in circulation, driven by the free thiol group at cysteine-34 (Cys34). This thiol acts as a potent nucleophile, scavenging superoxide and hydroxyl radicals and neutralizing peroxynitrite (Anraku et al., 2013).
- Recombinant advantage: Plasma-derived HSA is often oxidized, with only approximately 60% free thiol content. In contrast, high-quality rHSA (e.g., Optibumin®) can retain greater than 99% free thiol, resulting in significantly improved ROS
scavenging capacity compared to serum-derived equivalents (Anraku et al., 2013).
Metal Sequestration and Detoxification
Basal media are supplemented with transition metals such as copper and iron that are essential for metabolism, but problematic when unbound, as they catalyze Fenton reactions that generate hydroxyl radicals.
- The albumin chelate: P188 has no metal-binding capacity. Albumin binds copper and nickel at the N-terminal DAHK sequence with very high affinity affinity and possesses additional sites for zinc and iron (Linder, 2016).
- Albumin’s impact: By sequestering these ions, rHSA shuts down Fenton chemistry in bulk media while still delivering metals to cells via receptor-mediated uptake, preventing “trace metal toxicity” common in chemically defined processes (Zhu et al., 2012).
- P188 susceptibility: P188 itself is highly susceptible to oxidative degradation through Fenton-like reactions triggered by trace transition metals and peroxides. This degradation leads to surfactant loss and compromises product stability in biopharmaceutical formulations (Zegota, et al., 2025).
The Regulatory and Economic Considerations
Historically, albumin use declined due to viral contamination, including HIV and Hepatitis. As well as TSE/BSE risks associated with serum. Early recombinant approaches, such as yeast-based expression, proved cost-prohibitive for upstream bioprocessing. InVitria’s non-mammalian, non-microbial platform removes these barriers.
Scalability
InVitria’s production platform enables large-scale, cost-effective manufacture of recombinant human serum albumin suitable for bulk media supplementation. This approach overcomes the economic limitations associated with fermentation-based recombinant proteins.
Safety Profile
InVitria’s rHSA is animal-origin-free (AOF) and blood-free.
- Viral safety: The material is animal-origin-free and blood-free. The production system does not propagate mammalian viruses, eliminating risks associated with serum-derived albumin and reducing downstream viral clearance burden (He et al., 2011).
- Regulatory alignment: This material aligns with FDA and EMA directives to remove animal-derived materials from manufacturing while avoiding the impurity profile of industrial polymers.
Performance Data: Productivity and Quality
Replacing P188 with rHSA yields measurable process improvements beyond simple viability maintenance.
CHO Cell Growth and Titer
In comparative studies of CHO-K1 cell lines expressing monoclonal antibodies, supplementation with InVitria’s rHSA demonstrated clear superiority over P188-supplemented media.
- IVCC impact: rHSA supplementation increased the integral of viable cell concentration (IVCC) by approximately 50%.
- Titer impact: Volumetric antibody productivity increased by 92% to 140% compared to standard chemically defined media.
- Mechanism: The data suggests that by alleviating oxidative stress and optimizing lipid/metal delivery, rHSA allows cells to redirect metabolic energy from stress response to protein synthesis (Pettit et al., 2020).
Downstream T-Cell Washing
In cell therapy, P188 is often cytotoxic to sensitive primary T-cells.
- Viability retention: During high-shear washing steps, buffers containing rHSA retained more than 80% viable T-cells, whereas standard buffers resulted in significant losses. Albumin stabilizes the cell membrane during centrifugation without the membrane-fluidizing toxicity of P188 (Stathos et al., 2024).
Key Functional Differences: P188 vs. rHSA
From Synthetic Surrogates to Biological Function
The current challenges associated with poloxamer 188, the “Poloxamer Crisis”, reflect the limitations of synthetic surrogates in modern high-density bioprocessing. While P188 served as a transitional solution away from serum, its inherent variability and one-dimensional function now represent a process risk.
Recombinant human serum albumin, enabled by the scalability of InVitria’s non-mammalian expression system, restores the multi-functional support of biology without the safety risks of serum. It is not merely a shear protectant; it is a metabolic enhancer and a stabilizer of interfacial and redox environments. For biomanufacturers seeking higher productivity, greater robustness, and reduced supply chain risk, rHSA represents a technically superior alternative.
{
"@context": "https://schema.org",
"@type": "TechArticle",
"headline": "Beyond Synthetic Surfactants: The Case for Recombinant Human Serum Albumin (rHSA) as an Ideal Alternative to Poloxamer 188",
"description": "Technical article examining the limitations of poloxamer 188 in mammalian cell culture and the functional differences of recombinant human serum albumin (rHSA) related to shear protection, oxidative stress, and metal-mediated instability in upstream biomanufacturing.",
"inLanguage": "en",
"keywords": "poloxamer 188, P188, recombinant human serum albumin (rHSA), animal-origin-free, CHO cells, mammalian cell culture, shear protection, foam stability, lot-to-lot variability, oxidative stress, metal sequestration, upstream bioprocessing",
"articleSection": [
"Introduction",
"The Poloxamer 188 Crisis: Synthetic Variability and Risk",
"Mechanism of Action: The Biological Superiority of rHSA",
"Regulatory and Economic Considerations",
"Performance Data: Productivity and Quality",
"Conclusion",
"References"
],
"author": [
{
"@type": "Person",
"name": "Marcus Curl",
"affiliation": {
"@type": "Organization",
"name": "InVitria"
}
}
],
"publisher": {
"@type": "Organization",
"name": "InVitria",
"url": "https://invitria.com/",
"logo": {
"@type": "ImageObject",
"url": "https://invitria.com/path-to-logo.png"
}
},
"datePublished": "Unknown",
"copyrightYear": "2026",
"mainEntityOfPage": "https://invitria.com/resources//",
"url": "https://invitria.com/resources//",
"contentUrl": "https://invitria.com/resources/.pdf",
"about": [
{ "@type": "Thing", "name": "Shear protection in mammalian cell culture" },
{ "@type": "Thing", "name": "Lot-to-lot variability of synthetic surfactants" }
],
"mentions": [
{
"@type": "Product",
"name": "Optibumin®",
"brand": {
"@type": "Brand",
"name": "InVitria"
}
}
]
}
The following content is gated. Please, subscribe to open access to it.
Footnotes
References
- Anraku, M., Takeuchi, K., Watanabe, H., Kadowaki, D., Kitamura, K., Tomita, K., Kuniyasu, A., Suenaga, A., Maruyama, T., & Otagiri, M. (2011) Quantitative analysis of cysteine-34 on the antioxidative properties of human serum albumin in hemodialysis patients. Journal of Pharmaceutical Sciences, 100(9), 3968–3976. https://doi.org/10.1002/jps.22571
- Bandyopadhyay, A., Kosanam, H., Yang, R.-S., Gupta, B., Naralakattu, N., Pakhale, S., Kress, J., Richardson, D., & Ly, J. (2022). Low-molecular-weight impurity in poloxamer 188 responsible for atypical cell culture performance for mAb production. Journal of Biotechnology, 351, 13–22. https://doi.org/10.1016/j.jbiotec.2022.04.008
- Francis, G. L. (2010). Albumin and mammalian cell culture: Implications for biotechnology applications. Cytotechnology, 62(1), 1–16. https://doi.org/10.1007/s10616-010-9263-3
- Garcia-Briones, M., & Chalmers, J. J. (1992). Cell-bubble interactions: Mechanisms of suspended cell damage. Annals of the New York Academy of Sciences, 665, 219–229. https://doi.org/10.1111/j.1749-6632.1992.tb42586.x
- He, Y., Ning, T., Xie, T., & Yang, D. (2011). Large-scale production of functional human serum albumin from transgenic rice seeds. Proceedings of the National Academy of Sciences of the United States of America, 108(47), 19078–19083. https://doi.org/10.1073/pnas.1109736108
- Linder, M. C. (2016). Ceruloplasmin and other copper binding components of blood plasma and their functions: An update. Metallomics, 8(9), 887–905. https://doi.org/10.1039/c6mt00103c
- Peng, H., Ali, A., Lanan, M., Hughes, E., Wiltberger, K., Guan, B., Prajapati, S., & Hu, W. (2016). Mechanism investigation for poloxamer 188 raw material variation in cell culture. Biotechnology Progress, 32(3), 767–775. ttps://doi.org/10.1002/btpr.2268
- Peng, H., Hall, K. M., Clayton, B. L., Wittberger, K., Hu, W., Hughes, E., Kane, J., Ney, R., & Ryll, T. (2014). Development of small scale cell culture models for screening poloxamer 188 lot-to-lot variation. Biotechnology Progress 30 https://doi.org/10.1002/btpr.1967
- Pettit, S., Santos, M. A., Tanner, T., & Huang, N. (2020). Recombinant human serum albumin expressed in plants improves the productivity and growth kinetics of CHO [Poster]. Cell & Gene. InVitria. https://www.cellandgene.com/doc/recombinant-human-serum-albumin-expressed-in-plants-improves-the-productivity-and-growth-kinetics-of-cho-0001
- Stathos, M., et al. (2024). Maintaining viable cell count in primary T cells during cell washing using recombinant human serum albumin (rHSA) [Application note]. InVitria, Inc. https://invitria.com/resources/optibumin-tcell-washing/
- Zegota, M. M., Schöneich, C., & Hawe, A. (2025). Impact of biological buffers and chelators on Fenton-like reactions in the context of poloxamer 188 degradation. European Journal of Pharmaceutics and Biopharmaceutics, 216, 114855. https://doi.org/10.1016/j.ejpb.2025.114855
- Zhu, J., Wooh, J. W., Hou, J. C., Hughes, B. S., Gray, P. P., & Munro, T. P. (2012). Recombinant human albumin supports single cell cloning of CHO cells in chemically defined media. Biotechnology Progress, 28(3), 887–891. https://doi.org/10.1002/btpr.1549