- Home
- ITS-Animal-Free™ Supplementation for iPSC Expansion and Pluripotency: A Platform Approach for Serum-Free Cell Expansion
ITS-Animal-Free™ Supplementation for iPSC Expansion and Pluripotency: A Platform Approach for Serum-Free Cell Expansion
Published on 1 July 2025
Application Note
Authors: Mark Stathos, PhD, Product Applications Scientist & Andrew Hamann, PhD, Product Applications Scientist
InVitria, Inc., USA
EXECUTIVE SUMMARY
ITS Animal-Free™ , InVitria’s 100x cocktail of Insulin, Transferrin, and Selenium that is completely animal-origin-free, is essential for culture of all mammalian cell types including iPSCs. ITS AF can be conveniently supplemented into custom media formulations to replace the core components of serum. This reduces variability, eliminates safety and regulatory hurdles, and allows users to focus on the finer points when optimizing custom media formulations. Here we show that InVitria® ITS AF enhances iPSC proliferation and maintains pluripotency comparably to the leading competitors.
Key Findings
- Enables robust iPSC proliferation in serum-free systems
- Maintains ≥85% Oct4+/Sox2+/Nanog+ pluripotent population
- Performs equivalently to leading ITS products without optimization
- Animal-origin-free and blood-free, supporting GMP-ready workflows
INTRODUCTION
Since the discovery of induced pluripotent stem cells (iPSCs) (Takahashi and Yamanaka, 2006), scientists have sought to better define culture conditions for this highly sensitive cell type to enable facile culture, maintenance of pluripotency, and controlled reproducible differentiation into many target cell types (Villa-Diaz et al., 2013). The earliest stem cell protocols required feeder cells and serum (Thomson et al., 1998). The feeder cells initially employed introduced significant variability and required complex workflows (Li et al. 2017) and serum inclusion was also untenable due to the risk of xenogeneic contamination, regulatory concerns, and variability due to undefined components such as growth factors that induced unwanted differentiation (Lyra-Leite et al., 2022).
To address this, members of James Thomson’s lab developed the fully defined minimal essential iPSC media, Essential 8™ (E8) (Chen et al. 2011). This formulation consists of just eight ingredients including a base media (DMEM/F12), a buffering agent (sodium bicarbonate), an antioxidant (vitamin C), two growth factors (TGFβ and FGF2) as well as Insulin, Transferrin, and Selenium (ITS). It is no coincidence that ITS was included in E8 media as these components have all long been known to play indispensable roles in cell culture and to act in concert as a serum replacement (Guilbert and Iscove, 1976).
Insulin has been demonstrated to be a universal growth factor in human cell culture (Barnes and Sato, 1980). It activates cell signaling pathways that drive cell cycle progression even in cells that are arrested in G0/G1 due to nutrient limitation and plays important roles in cell metabolism (Straus, 1981).
Transferrin is the principle physiological delivery vehicle of iron to cells. Iron is essential for cellular respiration (Luck and Mason, 2015) and is a crucial co-factor for enzymes involved in DNA synthesis (Laskey, 1988). Furthermore, transferrin delivers iron through receptor mediated endocytosis, meaning cells can regulate its intake to minimize iron-induced oxidative stress.
Selenium is a trace element that is incorporated into antioxidant enzymes like glutathione peroxidase and thioredoxin reductase in the form of the noncanonical amino acid selenocysteine. It is essential for moderating oxidative stress and plays a critical role in cell cycle progression (Zeng, 2009).
InVitria’s ITS-AF is a pre-formulated, optimized, animal-origin-free serum replacement cocktail conveniently available as a 100x liquid solution. This format not only enables serum replacement for all cell types but also facilitates custom media formulation which is ubiquitous among clinical iPSC research groups.
Here we showcase directly the critical role of ITS in proliferation and maintenance of pluripotency in iPSCs. Due to the essential nature of ITS components, these results are applicable to any human cell type.
RESULTS AND DISCUSSION
ITS Enhances iPSC Proliferation
To assess how ITS supplementation affects iPSC proliferation, researchers first created an in-house “E5” medium—identical to commercial E8 but lacking ITS. InVitria ITS-AF or a leading competitor’s ITS was then added back into E5 at final concentrations ranging from 1x to 5x. (For reference, standard E8 media contains 2x ITS.)
Human iPSCs previously maintained in E8 were plated in 96-well plates with 10 µM Y-27632 ROCK inhibitor. After 24 hours, cells were switched to one of the following conditions: E5 alone, E5 with InVitria ITS-AF, E5 with competitor ITS, or complete E8. Cell proliferation was monitored every 2 hours over the next 48 hours using high-content imaging.
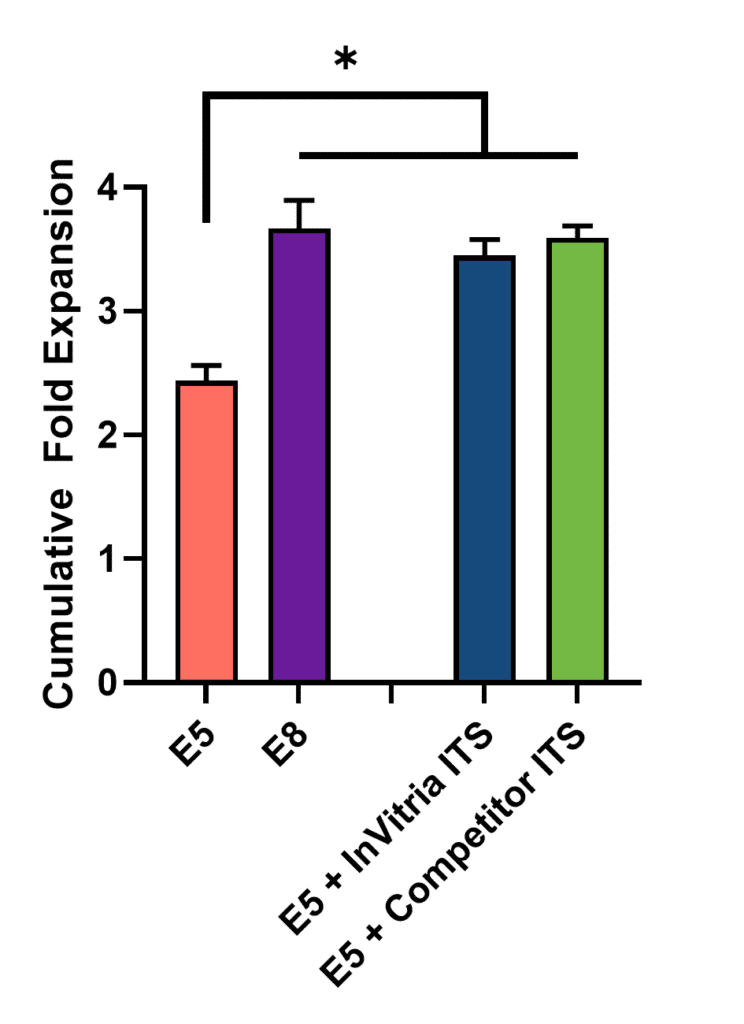
As shown in Figure 1, both InVitria ITS AF and competitor ITS supported robust expansion at 2x concentrations, equivalent to E8. Results at other concentrations (1x–5x) showed similar trends and are omitted for clarity. The consistency across doses indicates that InVitria ITS AF does not require titration or optimization during media formulation.
All media conditions containing ITS supported significantly greater iPSC proliferation compared to the E5 control lacking ITS. This difference is likely due to the absence of insulin (which supports metabolic activity), transferrin (which delivers iron for respiration and DNA synthesis), and selenium (which protects against oxidative stress).
No significant differences were observed between the InVitria ITS AF and the competitor formulations at any tested concentration, indicating comparable performance. These findings suggest that ITS-AF does not require further optimization during media development. Given the essential roles of ITS components, it is expected that these benefits would become even more pronounced over extended culture periods.
ITS Maintains Pluripotency in iPSCs
To further characterize the effect of ITS on iPSC culture, the cells that had been expanded in different amounts of ITS were then analyzed by flow cytometry for the core pluripotency markers Oct4, Sox2, and Nanog which are essential for maintenance of iPSCs (Rodda, 2005) (Figure 2).
All conditions supplemented with ITS maintained at least 85% triple-positive cells, indicating high pluripotency. In contrast, the ITS-free control group showed only 55% pluripotency. The larger variability in this negative control is likely due to the limited number of viable cells available for analysis. The observed loss of pluripotency may be linked to disrupted iron homeostasis in the absence of transferrin (Han et al., 2018).
Taken together with the expansion data, this phenotyping result suggests that ITS is critical for maintaining proliferating cells as well as preventing iPSCs from spontaneously differentiating. Once again there were no significant differences between conditions that contained ITS suggesting that InVitria’s ITS-AF is comparable to the leading competitor at every dose evaluated. Furthermore, the lack of a dose response suggests that ITS does not need to be titrated which simplifies custom media formulation.

CONCLUSION
This dataset demonstrates that ITS is essential for maintaining both expansion and pluripotency of iPSC cultures. Without these critical components, cultures fail to propagate efficiently and begin to spontaneously differentiate. Additionally, InVitria ITS-AF was shown perform as well as a leading competitor ITS product while also being completely defined and animal free. This makes InVitria ITS-AF a strong foundation on which to build custom media formulations that are high performance, chemically defined, and animal free.
MATERIALS AND METHODS
Human iPSCs maintained in Essential 8™ media (ThermoFisher Scientific) were dissociated using Accutase® (ThermoFisher Scientific) and seeded into 96-well plates at 10,000 cells per well. Each well was coated with 0.5 µg/cm² N-terminal truncated Vitronectin™ (ThermoFisher Scientific) and supplemented with 10 µM Y-27632 ROCK inhibitor.
Cells were cultured in one of the following conditions:
- E5 media (homemade Essential 8 without ITS)
- E5 supplemented with 1–5x InVitria ITS AF™ or competitor ITS
- Complete commercial Essential 8
Plates were incubated on an Incucyte SX5 system (Sartorius), and proliferation was monitored via confluence measurements taken every 2 hours. After 24 hours, the media was changed to remove ROCK inhibitor, and monitoring continued for an additional 24 hours.
At 48 hours, cells were harvested using Accutase® and stained for flow cytometry.
- Viability was assessed using Zombie Aqua™ fixable dye (BioLegend).
- Cells were fixed and permeabilized with the FOXP3 eBioscience™ buffer set (ThermoFisher Scientific).
- Cells were stained with pluripotency markers:
- Oct4 Alexa Fluor® 647
- Sox2 Alexa Fluor 488
- Nanog Alexa Fluor 594 (BioLegend antibody catalog)
Samples were analyzed using a CytoFLEX LX™ flow cytometer (Beckman Coulter).
Pluripotency was defined as the percentage of live cells triple-positive for Oct4, Sox2, and Nanog.
Featured Solution

ITS Animal-Free – Recombinant Insulin, Transferrin, and Selenium Supplement
ITS Animal-Free™ (ITS AF) is a chemically defined, animal-origin-free 100x liquid supplement designed to replace serum in cell culture. It supports robust proliferation and maintains pluripotency in iPSCs and other mammalian cell types. ITS AF eliminates the variability and regulatory concerns associated with serum-derived inputs, enabling more consistent, compliant, and scalable workflows in cell and gene therapy, regenerative medicine, and vaccine development.
Download the Full Application Note

The following content is gated. Please, subscribe to open access to it.
Footnotes
References
- Takahashi, K., & Yamanaka, S. (2006). Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell, 126(4), 663–676. https://doi.org/10.1016/j.cell.2006.07.024
- Villa-Diaz, L. G., Ross, A. M., Lahann, J., & Krebsbach, P. H. (2013). Concise review: The evolution of human pluripotent stem cell culture: From feeder cells to synthetic coatings. Stem Cells, 31(1), 1–7. https://doi.org/10.1002/stem.1190
- Thomson, J. A., Itskovitz-Eldor, J., Shapiro, S. S., Waknitz, M. A., Swiergiel, J. J., Marshall, V. S., & Jones, J. M. (1998). Embryonic stem cell lines derived from human blastocysts. Science, 282(5391), 1145–1147. https://doi.org/10.1126/science.282.5391.1145
- Li, P., Wang, S., Zhan, L., He, X., Chi, G., Lv, S., Xu, Z., Xia, Y., Teng, S., Li, L., & Li, Y. (2017). Efficient feeder cells preparation system for large-scale preparation and application of induced pluripotent stem cells. Scientific Reports, 7, 12266. https://doi.org/10.1038/s41598-017-10428-5
- Lyra-Leite, D. M., Gutiérrez-Gutiérrez, Ó., Wang, M., Zhou, Y., Cyganek, L., & Burridge, P. W. (2022). A review of protocols for human iPSC culture, cardiac differentiation, subtype-specification, maturation, and direct reprogramming. STAR Protocols, 3(3), 101560. https://doi.org/10.1016/j.xpro.2022.101560
- Chen, G., Gulbranson, D. R., Hou, Z., Bolin, J. M., Ruotti, V., Probasco, M. D., Smuga-Otto, K., Howden, S. E., Diol, N. R., Propson, N. E., & Thomson, J. A. (2011). Chemically defined conditions for human iPSC derivation and culture. Nature Methods, 8(5), 424–429. https://doi.org/10.1038/nmeth.1593
- Guilbert, L. J., & Iscove, N. N. (1976). Partial replacement of serum by transferrin, albumin and lecithin in haemopoietic cell cultures. Nature, 263(5574), 594–595. https://doi.org/10.1038/263594a0
- Barnes, D., & Sato, G. (1980). Serum-free cell culture: A unifying approach. Cell, 22(3), 649–655. https://doi.org/10.1016/0092-8674(80)90540-1
- Straus, D. S. (1981). Effects of insulin on cellular growth and proliferation. Life Sciences, 29(21), 2131–2139. https://doi.org/10.1016/0024-3205(81)90482-3
- Luck, A. N., & Mason, A. B. (2012). Transferrin-mediated cellular iron delivery. Current Topics in Membranes, 69, 3–35. https://doi.org/10.1016/B978-0-12-394390-3.00001-X
- Laskey, J., Webb, I., Schulman, H. M., & Ponka, P. (1988). Evidence that transferrin supports cell proliferation by supplying iron for DNA synthesis. Experimental Cell Research, 176(1), 87–95. https://doi.org/10.1016/0014-4827(88)90123-1
- Zeng, H. (2009). Selenium as an essential micronutrient: Roles in cell cycle and apoptosis. Molecules, 14(3), 1263–1278. https://doi.org/10.3390/molecules14031263
- Rodda, D. J., Chew, J. L., Lim, L. H., Loh, Y. H., Wang, B., Ng, H. H., & Robson, P. (2005). Transcriptional regulation of Nanog by OCT4 and SOX2. Journal of Biological Chemistry, 280(26), 24731–24737. https://doi.org/10.1074/jbc.M502573200
- Han, Z., Yu, Y., Xu, J., Bao, Z., Xu, Z., Hu, J., Yu, M., Bamba, D., Ma, W., Ding, F., Zhang, L., Jin, M., Yan, G., Huang, Q., Wang, X., Hua, B., Yang, F., Li, Y., Lei, L., … Cai, B. (2019). Iron homeostasis determines fate of human pluripotent stem cells via glycerophospholipids-epigenetic circuit. Stem Cells, 37(4), 489–503. https://doi.org/10.1002/stem.2967