- Home
- Preserving Lentiviral Titer During Sterile Filtration Using Recombinant Albumin
Preserving Lentiviral Titer During Sterile Filtration Using Recombinant Albumin
Published on 31 March 2026
Application Note
Author(s): Mark Stathos, PhD, Applications Scientist, Wren Michaels, PhD, Molecular Biology Scientist, Jacob Weber, PhD, Vice President of Process Development
InVitria, Inc., USA
EXECUTIVE SUMMARY
Lentivirus production is central to CAR-T manufacturing, and is even more critical with the emergence of in vivo CAR-T approaches. This puts increased emphasis on the need for high-yield lentivirus manufacturing. However, lentiviruses are inherently fragile, and infectious titer is lost at every step in manufacturing. The shift towards suspension systems, and chemically defined, serum-free media further increases stress on lentiviral vectors (LVVs), negatively impacting stability, recovery and yield.
In downstream processing, final sterile filtration is a particularly egregious pain point despite the use of low flow rates and low protein binding filters. In this application note, we show that incorporation of an excipient-grade recombinant albumin, Optibumin® by InVitria, in pre-filtration formulations, subsequent line-flushing, and pre-coating filters is a cost-effective and straightforward way to preserve infectious lentivirus titer during filtration and is suitable for in vivo CAR-T applications due to Optibumin’s excipient status.
Key highlights
- ~75% genomic titer retained vs ~19% without Optibumin
- ~49% infectious titer retained vs ~17% without
- ~3-fold increase in infectious titer recovery
- Effective filter passivation with 1% Optibumin in 5 minutes
- Maintains expected infectious-to-genomic titer ratios
INTRODUCTION
Lentiviruses are RNA retroviruses which have been engineered to serve as powerful genome editing tools for both basic research and advanced therapies (Levine et al., 2006). These viruses can stably integrate genes of interest such as chimeric antigen receptors (CARs), across a range of clinically relevant cell types including such as activated T cells (Porter et al., 2011), NK cells (Portillo et al., 2021), HSCs (Srivastava et al., 2024), and iPSCs (Nethercott et al., 2013), whether they are proliferating or quiescent (Sakuma et al., 2012).
Recent developments in in vivo CAR-T therapies, initially developed by Fred Hutchinson Cancer Research Center (Smith et al. 2017), and further advanced by groups such as Interius (Andorko et al., 2025), Umoja (Michels et al., 2023) and Azalea (Nyberg et al., 2026) may now enable advanced therapies with shorter, simpler, less expensive and scalable manufacturing workflows than have been possible with current processes involving ex vivo cell expansion. This makes reliable lentivirus production more important than ever for progress in cancer therapy.
Due to their structure, lentiviral vectors (LVVs) are notoriously fragile. LVVs are enveloped viruses coated in a lipid bilayer derived from their host cell during viral secretion, and this envelope is essential to their infectious function. Because of this coating, LVVs may loosely be thought of as small cells, and they are vulnerable to many of the same stressors that are harmful to cells including high salt concentration (Shi et al., 2022), highly acidic or basic pH (Boudeffa et al., 2019), high shear (Perry et al., 2023), and elevated temperature (Carmo et al., 2009).
Harvest, clarification, purification, and filtration, which are essential unit operations in LVV manufacturing, inevitably subject viral samples to many of these very stressors, meaning substantial amounts of this hard-earned and expensive infectious titer are lost throughout downstream processing (Moreira et al., 2021; Barbieri et al., 2026). While every unit operation is problematic, final sterile filtration of the 0.1-micron diameter virus through a 0.2 micron filter is a significant pain point causing infectious titer losses of greater than 30% with losses being even greater at high viral concentration (Vogel et al., 2024). The main mechanisms for this loss are nonspecific adsorption of the virus to the filter and tearing of the viral envelope due to shear and transmembrane pressure.
To address these issues, LVV processes generally utilize low binding membrane chemistries such as polyether sulfone (PES) or polyvinylidene fluoride (PVDF) to minimize adsorption and carefully control flux over the filter while also attempting to reduce processing times (Vogel et al., 2024). However, significant losses are still observed with PES and PVDF and there is generally a tradeoff between processing time and stress due to shear and transmembrane pressure at higher flow rates.
To further improve infectious LVV titer, coating filters with carrier proteins such as albumin has been shown to be an effective strategy (Bandeira et al., 2012; Cytiva 2024) and albumin is also widely used as a stabilizer in LVV DSP (Carmo et al., 2009; Moreira et al., 2021; Shi et al., 2022; Perry et al., 2023). Albumin acts as a shear protectant, prevents aggregation of viral particles, and non-specifically coats surfaces such as filters to reduce LVV adsorption. Furthermore, the use of InVitria recombinant albumin such as Optibumin and Exbumin to stabilize VSV-G pseudotyped envelope viruses specifically has precedent based on the formulation of the Ervebo® Ebola Vaccine developed by Merck (EMA 606159,2019; Burke and Volkin US Patent 6,210,683).
In this application note, we quantify the benefit of using Optibumin 25, a cGMP animal-free excipient grade recombinant albumin, to pre-coat sterile filters and tubing to maintain infectious LVV titer.
RESULTS AND DISCUSSION
To assess the effect of albumin pre-treatment on sterile filters, 0.2 micron PES filters were pre-coated with formulations comprised of Hanks’ balanced salt solution (HBSS) with or without the addition of 1% (m/v) Optibumin 25 for 5 minutes, the buffer was then slowly pumped through the 30 mm diameter 0.22 micron PES filter at a flow rate of 2 mL per minute. The buffer was then immediately followed by a sample containing commercially available purified VSV-pseudotyped GFP lentivirus diluted in the same buffer at the same flow rate. The tubing and filter were replaced between samples. An identical unfiltered sample was also prepared as a control.
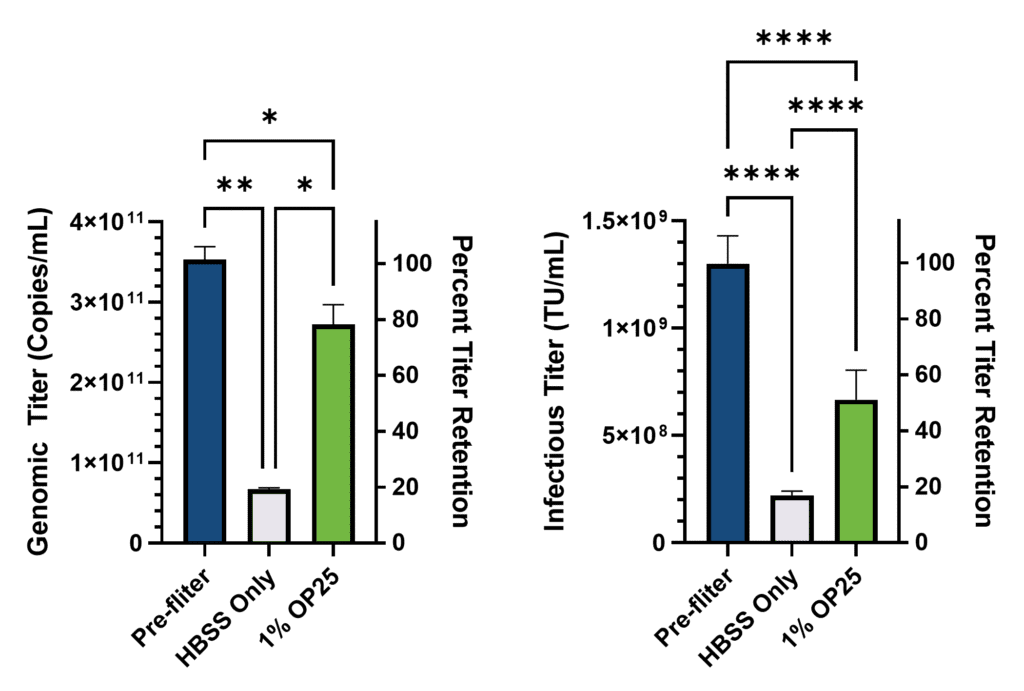
After filtration, absolute genomic titer was quantified by RT-qPCR against a standard curve of RNA of known concentration. The titer of the initial concentrated sample was back-calculated based on the dilution series and expressed relative to the original sample, which was supplied at 1.00 × 10⁹ TU/mL (Figure 1).
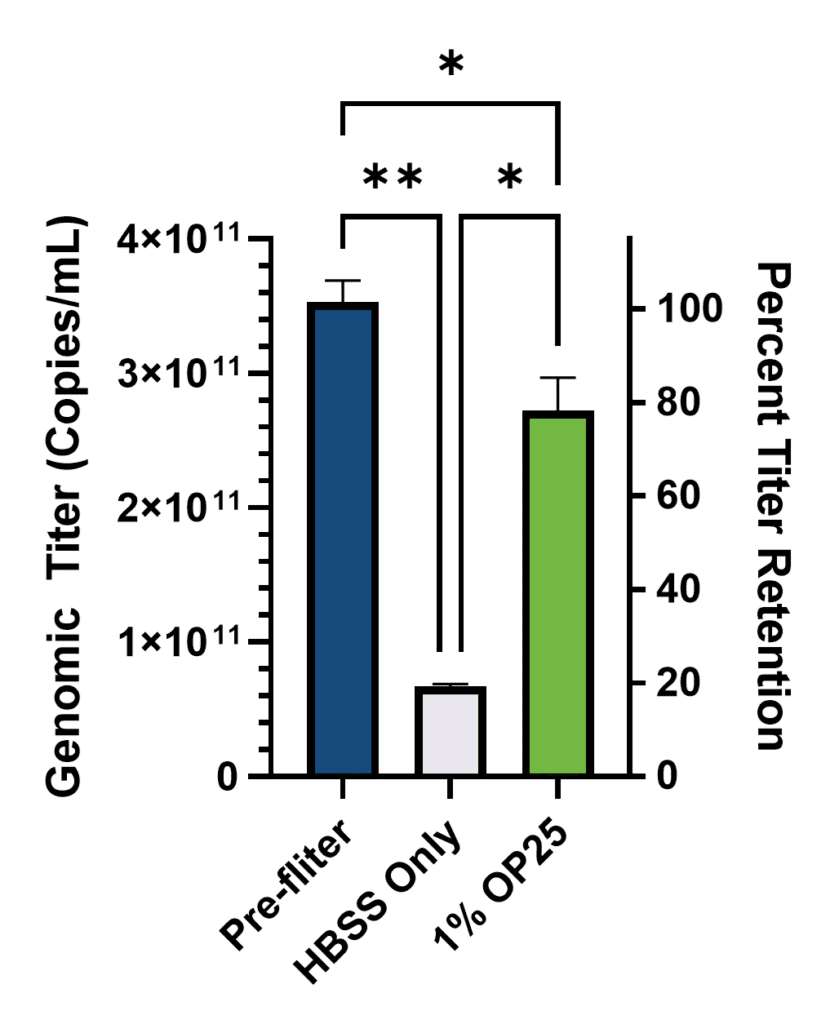
As expected, significant losses in genomic titer were observed after filtration regardless of treatment. But importantly, the inclusion of albumin significantly reduced these losses. In the buffer only condition, 81% of the genomic titer was lost due to filtration compared to only 25% lost in the Optibumin group. Alternatively, only 19% of the genomic titer was retained after filtration in the sample with only buffer while 75% percent of the titer was retained in the Optibumin group, which is a greater than threefold increase. Notably, the genomic titer was approximately 300 times the labeled infectious titer which aligns with expectations of a 100:1 to 1000:1 physical to infectious particle ratio for LVVs (Stibbs et al., 2024).
To corroborate the genomic titer benefits observed by RT-qPCR, infectious titer, which is arguably the most critical metric for cell and gene therapy applications, was measured using an image-based approach to observe the percentage of GFP positive HT-1080 cells 72 hours after transduction (Figure 2).
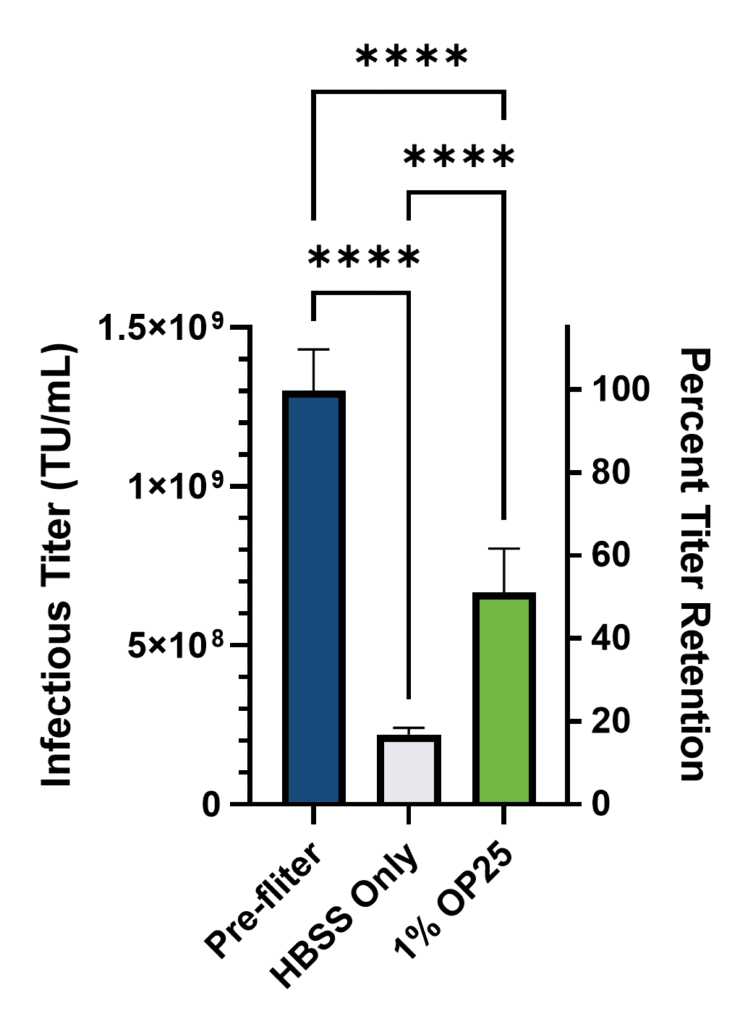
Once again, expected substantial losses in titer were observed post filtration, but inclusion of 1% albumin significantly reduced these losses. Specifically, 83% of the infectious titer was lost in the group with only buffer (17% retained) while only 51% was lost in the Optibumin group (49% retained). Critically, this is nearly a 3-fold increase in retained infectious titer due to Optibumin.
Taken together, these results demonstrate that pre-treating filters with 1% Optibumin for as little as five minutes significantly increases both genomic and infectious final titer without a substantial impact on processing time or infectious titer to genomic titer ratio (0.36% which is in line with typical values of 0.1 to 1%). Additionally, because titer loss tends to be greater at higher viral concentrations (Merten et al., 2015; Gernier et al., 2008; Vogel et al., 2024), we expect that these benefits would scale effectively.
If the threefold increase in retained infectious titer is assumed to scale to industrial processes, and the cost of goods for lentivirus production per dose of CAR-T cell therapy is about $100,000 per dose in the earliest phase of clinical trials (Comisel et al., 2021), then addition of albumin could save up to $67,000 per dose.
CONCLUSION
Utilizing a scalable filtration model representative of industrial shear parameters, we demonstrate that passivating 0.22-micron PES membranes with recombinant, animal-free Optibumin significantly enhances both genomic and infectious LVV recovery. Furthermore, the incorporation of Optibumin into pre-filtration formulation and subsequent line and filter flushing protocols effectively mitigates non-specific adsorptive losses throughout the downstream processing DSP workflow. Integrating Optibumin provides a robust, cost-effective, and chemically defined strategy for maximizing final infectious titers while maintaining a defined, animal-free manufacturing environment.
MATERIALS & METHODS
Virus Filtration
Aliquots of purified GFP lentivirus samples (VectorBuilder®, LVMP(VB010000-9492agg)) at 1.00 × 10⁹ TU/mL were diluted in Hanks’ balanced salt solution with or without 1% (m/v) Optibumin and stored on ice when not in use. A MasterFlex® L/S® Peristaltic Pump (Cole-Palmer) with an Easy-Load® II pump head was affixed with 10 mm diameter MasterFlex L/S platinum-cured silicone tubing with a 30 mm diameter 0.2 micron PES filter attached at one end. The tubing was pre-equilibrated with virus-free buffer corresponding to the test sample for five minutes to enable adsorption of albumin to the filter surface. After the equilibration period, the samples were continuously pumped at 2 mL per minute through the tubing and filter and the filtrate retained for titer measurements. An unfiltered virus sample was also prepared and retained for titer measurement. Fresh tubing was used for each sample. The 2 mL per minute flow rate was chosen such that it would scale approximately to industrially relevant shear stress and transmembrane pressure.
Genomic Titer Quantitation
Lentiviral genomic titer was measured by harvesting viral supernatants. Viral RNA was extracted using the NucleoSpin® RNA Virus Kit (Takara Bio, #740956). Extracted RNA was treated with DNase I to remove any residual plasmid DNA. Purified viral RNA was reverse transcribed and amplified using the Lenti-X® qRT-PCR Titration Kit (Takara Bio, #631236) for quantification using SYBR® Green (Thermo Fisher Scientific) on the QuantStudio™ 3 System (Applied Biosystems, a Thermo Fisher brand). Samples were run in serial dilutions to determine threshold cycle (Ct) values. Ct values were fit to a standard curve of control template RNA with known copy numbers and used to calculate copies per mL. Dilutions outside of the standard curve, or with multiple melt curve peaks, were discarded from analysis.
Infectious Titer Quantitation
HT-1080 cells were seeded at 7000 cells per mL in a 96-well plate in DMEM with 10% FBS and returned to the incubator to allow adhesion. While the cells were adhering, filtered lentivirus samples were serially diluted in DMEM with 10% FBS supplemented with Synperonic® F108 (Croda International). Four hours after plating the cells, the media was exchanged with the diluted virus samples, and the cells were spinoculated at 900 x g for 30 minutes at room temperature before being returned to the incubator. 16 hours later, the media was replaced with fresh DMEM with 10% FBS and the cells were monitored for GFP expression using an Incucyte® SX5 live cell imaging system. At 72 hours post transduction, the percentage of GFP positive cells was recorded. Virus dilutions for which the non-filtered control sample had GFP expression within the linear range of the assay (2.5% to 20% GFP positive) were used to compute the titer using the following equation:
Titer=(F*C/V)*D
Where
F = frequency of GFP+ cells (%GFP+ cells/100)
C = cell number per well at transduction (7,000 cells)
V = volume per well in mL
D = lentivirus dilution factor
Featured Solution
Optibumin 25 – Recombinant Human Serum Albumin, 25% Solution – Animal-Origin-Free, GMP-Produced
Optibumin® 25 is a chemically defined, recombinant human serum albumin designed to improve lentiviral vector (LVV) stability and recovery during downstream processing. As demonstrated in this application note, pre-coating sterile filtration systems with 1% Optibumin significantly reduces titer loss by minimizing non-specific adsorption and protecting viral integrity under shear conditions.
Unlike plasma-derived albumin, Optibumin is free from stabilizers such as caprylate and N-acetyl tryptophan and avoids variability associated with blood-derived sourcing. This results in a consistent, animal-origin-free solution that integrates easily into existing workflows through filter pre-coating and line flushing.
Optibumin 25 is well-suited for gene therapy and cell therapy applications where maintaining infectious titer and process consistency is critical.
Ready to try Optibumin 25?
Get a free evaluation sample or request a quote for your project.
Preserving Lentiviral Titer Frequently Asked Questions (FAQs)
Lentivirus Filtration Performance
How Much Lentiviral Titer Is Lost During Sterile Filtration?
Without protective measures, sterile filtration of lentiviral vectors through 0.22-micron PES filters typically results in greater than 30% infectious titer loss, with losses being even greater at high viral concentration. In this application note, buffer-only filtration retained only ~19% genomic titer and ~17% infectious titer. Pre-treating the same filters with 1% Optibumin 25 recombinant human albumin recovered ~75% genomic titer and ~49% infectious titer — nearly a 3-fold improvement in retained infectious titer.
How Does Recombinant Albumin Protect Lentiviral Vectors During Filtration?
Recombinant albumin protects lentiviral vectors through three mechanisms: acting as a shear protectant during pumping and filtration, preventing aggregation of viral particles, and non-specifically coating filter membrane surfaces to reduce non-specific viral adsorption. Pre-coating 0.22-micron PES filters with 1% Optibumin 25 for as little as 5 minutes effectively passivates the membrane and significantly reduces titer loss without impacting processing time.
What Concentration of Optibumin 25 Is Used for Filter Pre-Treatment?
1% (m/v) Optibumin 25 in Hanks’ balanced salt solution (HBSS) was used to pre-coat 0.22-micron PES filters and tubing for 5 minutes prior to lentivirus filtration. This concentration was sufficient to passivate the filter membrane and significantly reduce non-specific viral adsorption during subsequent filtration at 2 mL/min flow rate.
What Types of Lentiviral Vectors Does Optibumin 25 Protect?
This application note specifically validates Optibumin 25 with VSV-G-pseudotyped GFP lentivirus, which is the most common envelope used in clinical lentiviral vector manufacturing for cell and gene therapy. The mechanism of albumin-mediated viral envelope stabilization is the same mechanism underlying Merck’s ERVEBO® Ebola vaccine, which uses recombinant albumin to stabilize VSV-G-pseudotyped viral particles. The protective effect is expected to extend to other enveloped viral vectors used in similar manufacturing workflows.
Regulatory and Manufacturing
Is Optibumin 25 Suitable for In Vivo CAR-T Applications?
Yes. Optibumin 25 is a chemically defined, animal-origin-free, GMP-produced excipient-grade recombinant human serum albumin produced via the ExpressTec platform. The same recombinant albumin platform is used in Merck’s ERVEBO Ebola vaccine (EMA/606159/2019), establishing regulatory precedent for recombinant albumin as an injectable excipient for VSV-G-pseudotyped envelope viruses. This makes Optibumin 25 suitable for both ex vivo and in vivo CAR-T cell therapy applications.
What Is the Cost Benefit of Using Albumin in Lentivirus Filtration?
Based on published cost-of-goods analysis showing approximately $100,000 per dose for lentivirus production in early-phase CAR-T clinical trials (Comisel et al., 2021), the 3-fold improvement in retained infectious titer demonstrated with 1% Optibumin 25 filter pre-treatment could represent up to $67,000 in savings per CAR-T dose if the laboratory-scale results scale to industrial manufacturing processes.
Can These Results Scale to Industrial Lentivirus Manufacturing?
The 2 mL per minute flow rate used in this study was chosen specifically to scale approximately to industrially relevant shear stress and transmembrane pressure. Because titer loss tends to be greater at higher viral concentrations (Merten et al., 2015; Gernier et al., 2008; Vogel et al., 2024), the protective benefit of Optibumin 25 pre-treatment is expected to scale effectively to industrial lentivirus manufacturing processes. Contact InVitria’s Process Development team to discuss application of Optibumin 25 in your specific manufacturing workflow.
Footnotes
REFERENCES
- Andorko, J. I., Michels, A., Nyberg, S., & colleagues. (2025). Targeted in vivo delivery of genetic medicines utilizing an engineered lentiviral vector platform results in CAR T and NK cell generation. Molecular Therapy. https://doi.org/10.1016/j.ymthe.2025.06.036
- Barbieri, E., & Heldt, C. L. (2026). Challenges and opportunities in lentivirus viral vector manufacturing for in vivo applications. Biomedicines, 14(2), 369. https://doi.org/10.3390/biomedicines14020369
- Boudeffa, D., Rols, M. P., & Teissié, J. (2020). Toward a scalable purification protocol of GaLV-TR–pseudotyped lentiviral vectors. Human Gene Therapy Methods, 31(3–4), 129–139. https://doi.org/10.1089/hgtb.2019.076
- Burke, C. W., & Volkin, D. B. (2001). Stabilized viral formulations (U.S. Patent No. 6,210,683 B1). U.S. Patent and Trademark Office. https://patents.google.com/patent/US6210683B1/en
- Carmo, M., Dias, J. D., Sampaio, P., & Alves, P. M. (2009). Stabilization of gammaretroviral and lentiviral vectors from production to gene transfer. Journal of Gene Medicine, 11(8), 670–679. https://doi.org/10.1002/jgm.1353
- Comisel, R. M., Faulkner, P., Butler, M., & Gagnon, P. (2021). Lentiviral vector bioprocess economics for cell and gene therapy commercialization. Biochemical Engineering Journal, 168, 107868. https://doi.org/10.1016/j.bej.2020.107868
- Cytiva. (2024). Primary clarification of lentiviral vectors: Scalable lentiviral vector clarification. https://www.cytivalifesciences.com/en/us/insights/lentiviral-vector-clarification
- European Medicines Agency. (2019). Ervebo (Ebola Zaire vaccine) assessment report (EMA/606159/2019). https://www.ema.europa.eu/en/medicines/human/EPAR/ervebo
- Gernier, S., Jaffrin, M. Y., & Ding, L. H. (2008). Analysing flux decline in dead-end filtration. Chemical Engineering Research and Design, 86(10), 1131–1138. https://doi.org/10.1016/j.cherd.2008.06.005
- Levine, B. L., Humeau, L. M., Boyer, J., MacGregor, R. R., Rebello, T., Lu, X., Slepushkin, V., Lemiale, F., Mascola, J. R., Bushman, F. D., Dropulic, B., & June, C. H. (2006). Gene transfer in humans using a conditionally replicating lentiviral vector. Proceedings of the National Academy of Sciences. https://doi.org/10.1073/pnas.0608138103
- Merten, O.-W., Hebben, M., & Bovolenta, C. (2016). Production of lentiviral vectors. Molecular Therapy—Methods & Clinical Development, 3, 16017. https://doi.org/10.1038/mtm.2016.17
- Michels, K. R., Sheih, A., Hernandez, S. A., Brandes, A. H., Parrilla, D., Irwin, B., Perez, A. M., Ting, H.-A., Nicolai, C. J., Gervasio, T., Shin, S., Pankau, M., Mouhonen, M., Freeman, J., Gould, S., Getto, R., Larson, R. P., Ryu, B. Y., Scharenberg, A. M., Sullivan, A. M., & Green, S. (2023). Preclinical proof of concept for VivoVec, a lentiviral-based platform for in vivo CAR T-cell engineering. Journal for ImmunoTherapy of Cancer, 11(3), e006292.
https://doi.org/10.1136/jitc-2022-006292 - Moreira, A. S., Cavaco, D. G., Faria, T. Q., Alves, P. M., Carrondo, M. J. T., & Peixoto, C. (2021). Advances in lentivirus purification. Biotechnology Journal, 16(1), e2000019.
https://doi.org/10.1002/biot.202000019 - Nethercott, H. E., Brickman, J. M., & Szilvassy, S. J. (2013). Derivation of Induced Pluripotent Stem Cells by Lentiviral Transduction. In Methods in Molecular Biology (Vol. 997, pp. 67–78). Humana Press. https://doi.org/10.1007/978-1-61779-201-4_6
- Nyberg, W. A., Bernard, P.-L., Ngo, W., Wang, C. H., Ark, J., Rothrock, A., Borgo, G. M., Kimmerly, G. R., Jung, J. H., Allen, V., Hamilton, J. R., Baldwin, A., Stickels, R., Wyman, S., Khan, A. H., Lang, S., Marsh, D., Almudhafar, N., Novick, C., Mortazavi, Y., Zhang, S., AbdElwakil, M. M., Sandoval, L. R., Hwang, S., Chu, S. N., Jung, H., Liu, C., Sharma, D., McCreary, T., Li, Z., Satpathy, A. T., Carnevale, J., Rutishauser, R. L., Cromer, M. K., Roybal, K. T., Dodgson, S. E., Doudna, J. A., Asokan, A., & Eyquem, J. (2026). In vivo site-specific engineering to reprogram T cells. Nature. https://doi.org/10.1038/s41586-026-10235-x
- Perry, C., Mujahid, N., Teakuechi, Y., & Rayat, A. C. M. E. (2024). Insights into product and process related challenges of lentiviral vector bioprocessing. Biotechnology and Bioengineering, 121(8), 2466–2481. https://doi.org/10.1002/bit.28498
- Porter, D. L., Levine, B. L., Kalos, M., Bagg, A., & June, C. H. (2011). Chimeric Antigen Receptor–Modified T cells in Chronic Lymphoid Leukemia. New England Journal of Medicine, 365(8), 725–733. https://doi.org/10.1056/NEJMoa1103849
- Portillo-Lara, R., Annabi, N., Abbas, A., Ahn, E. H., Tamayol, A., & Khademhosseini, A. (2021). Production of human CAR-NK cells with lentiviral vectors and functional assessment in vitro. STAR Protocols, 2(3), 100956. https://doi.org/10.1016/j.xpro.2021.100956
- Sakuma, T., Barry, M. A., & Ikeda, Y. (2012). Lentiviral vectors: Basic to translational. Biochemical Journal. https://doi.org/10.1042/bj20120146
- Shi, R., Jia, S., Liu, H., & Nie, H. (2022). Clinical grade lentiviral vector purification and quality control requirements. Journal of Separation Science, 45(12), 2093–2101.
https://doi.org/10.1002/jssc.202100937 - Smith, T. T., Stephan, S. B., Moffett, H. F., McKnight, L. E., Ji, W., Reiman, D., Bonagofski, E., Wohlfahrt, M. E., Pillai, S. P., Stephan, M. T., & Irvine, D. J. (2017). In situ programming of leukaemia-specific T cells using synthetic DNA nanocarriers. Nature Nanotechnology. https://doi.org/10.1038/nnano.2017.57
- Srivastava, A., Rangarajan, S., Chowdary, P., Ye, J., de la Fuente, B., Ozelo, M. C., & others. (2024). Lentiviral Gene Therapy with CD34+ Hematopoietic Cells for Hemophilia A. New England Journal of Medicine. https://doi.org/10.1056/NEJMoa2410597
- Stibbs, H. H., Lendon, K., McDonald, D., McLachlan, R., Tapping, R. I., & Schaffer, D. V. (2024). Lentiviral vector adsorption and recovery during downstream processing: Mechanisms and mitigation strategies. Molecular Therapy: Methods &
Clinical Development, 32, 101209. https://doi.org/10.1016/j.omtm.2024.101209 - Vogel, J. E., Terrao, M., Schwingal, S., Kapitza, L., Brigulla, D., Pirzas, V., Laux, H., & Brandt, T. (2024). High yield sterile filtration process for highly concentrated lentiviral vectors. Biotechnology Journal, 19(3), e2300348. https://doi.org/10.1002/biot.202300348