- Home
- Replacing the “Necessary Evil”: Recombinant Human Serum Albumin as a Chemically Stable Alternative to Polysorbates in Biopharmaceutical Formulation
Replacing the “Necessary Evil”: Recombinant Human Serum Albumin as a Chemically Stable Alternative to Polysorbates in Biopharmaceutical Formulation
Published on 10 March 2026
Article
Marcus Curl, VP of Product Applications
Reading time: 4 minutes
Share this article:

Polysorbate 20 and polysorbate 80 have long been treated as default stabilizers in biopharmaceutical formulations. They are effective surfactants and, for many years, have been viewed as a practical solution to interfacial stress and aggregation. Over time, however, a growing body of evidence has shown that these excipients are not chemically inert.
Polysorbates undergo oxidative degradation and enzymatic hydrolysis in the presence of trace host cell proteins, generating insoluble fatty acids and reactive byproducts that can compromise shelf life and, in some cases, raise immunogenicity concerns. As biologics move toward higher concentrations and longer storage requirements, these limitations are becoming harder to ignore. This article explores why polysorbates behave the way they do and why recombinant human serum albumin is increasingly being evaluated as a more stable alternative.

The Polysorbate Failure Mechanism: A Chemical Liability
Polysorbates have been long regarded as inert formulation aids. However, post-2015 literature has definitively reclassified them as chemically reactive liabilities with intrinsic failure modes that exacerbated by modern manufacturing processes.
Enzymatic Hydrolysis: The Silent Degradation
The dominant failure mechanism in monoclonal antibody (mAb) formulations is enzymatic hydrolysis. Despite rigorous downstream purification (Protein A, IEX), trace levels of host cell lipases often co-purify with the drug substance.
- Culprits: Early investigations implicated Phospholipase B-Like 2 (PLBL2), but subsequent work identified lysosomal phospholipase A2 (LPLA2) and lipoprotein lipase (LPL) as primary contributors to polysorbate degradation (Hall et al., 2016). These enzymes remain catalytically active even at refrigerated temperatures (2–8 °C) and at sub-ppm concentrations.
- Mechanism: Lipases cleave the ester bond between the fatty acid chain and the polyoxyethylene sorbitan core, releasing free fatty acids (FFAs). PS80 primarily yields oleic acid, while PS20 releases lauric, myristic, and palmitic acids (Kishore et al., 2011).

- The Particle Cliff: Because FFAs exhibit poor aqueous solubility, they accumulate until a solubility threshold is exceeded, resulting in sudden precipitation of crystalline particles. This “particle cliff” often manifests late in stability studies, resulting in catastrophic batch failures after extended storage (12-24 months) (Saggu et al., 2015).

Oxidative Degradation
Concurrently, the polyoxyethylene (POE) chains of polysorbates undergo auto-oxidation triggered by light, heat, or transition metals.
- Radical Propagation: This radical-mediated chain reaction generates hydroperoxides, which decompose into reactive aldehydes (formaldehyde, acetaldehyde) and ketones (Kerwin, 2008).
- Collateral Damage: Unlike hydrolysis, which primarily affects the surfactant, oxidative byproducts are reactive toward the therapeutic protein itself. Peroxides oxidize susceptible amino acid residues, like methionine and tryptophan, while aldehydes form Schiff bases with primary amines, promoting covalent protein aggregation (Wang et al., 2011).
The Clinical Consequence: Immunogenicity
The degradation of polysorbates is not merely a quality control specification issue; it represents a direct threat to patient safety via immunogenicity.
Particulates as Unintended Adjuvants
The human immune system is evolutionarily tuned to recognize particulate matter as a “danger signal.”
- Mechanism: Insoluble fatty acid particles formed by polysorbate degradation can act as unintended adjuvants. When therapeutic proteins adsorb onto the particle surfaces, they are presented to antigen-presenting cells (APCs) in a repetitive, virus-like array (Singh, 2011).
- Breaking Tolerance: This particulate presentation can break B-cell tolerance, triggering the production of Anti-Drug Antibodies (ADAs). The historical Eprex® (erythropoietin) case serves as the definitive cautionary tale: Reformulation removed of human serum albumin (HSA), which traditionally masked impurities and stabilized the protein, and introduced Polysorbate 80, which interacted with syringe components. This created excipient-driven micelles that functioned as an unintentional adjuvant, leading to a catastrophic loss of immune tolerance and the development of pure red cell aplasia (Boven et al., 2005).
Albumin as a Biological Stabilizer
Human serum albumin (HSA) is the body’s primary extracellular stabilizer, evolved to bind hydrophobic molecules and protect plasma proteins. Unlike polysorbates, which function solely through surface activity, albumin provides both chaperone-like stabilization and antioxidant protection.
The Chaperone Effect
Albumin functions as an extracellular chaperone, preventing protein aggregation through specific hydrophobic interactions.
- Surface Competition: Like polysorbates, albumin is amphiphilic and coats hydrophobic surfaces (air-liquid interfaces), preventing loss of the therapeutic protein from adsorption to these surfaces as well as degradation due to unfolding at these boundaries (Finn et al., 2012).
- Hydrophobic Shielding: Crucially, HSA possesses conserved hydrophobic binding pockets (Sudlow Sites I and II). When a therapeutic protein undergoes thermal or stress-induced partial unfolding, HSA binds reversibly to the exposed hydrophobic patches. This “capping” prevents the unfolded protein from associating with others to form aggregates, effectively arresting the nucleation of amyloid fibrils (Khan & Naeem, 2024).
Antioxidant Capacity: The Cys34 Thiol
While polysorbates are pro-oxidants (generating peroxides), HSA is a potent antioxidant.
- The Free Thiol: HSA contains a single free cysteine residue at position 34 (Cys34). This thiol group (-SH) constitutes the major reducing power of human plasma (Oettl & Stauber, 2007).
- Radical Scavenging: Cys34 actively scavenges Reactive Oxygen Species (ROS) such as hydrogen peroxide and superoxide. By intercepting these radicals, albumin acts as a “sacrificial antioxidant,” becoming oxidized itself (to sulfenic acid) to protect the high-value therapeutic cargo from oxidative damage (Taverna et al., 2013).
The Recombinant Advantage: InVitria’s rHSA
Historically, the use of HSA was limited by the risks of plasma-derived sources (viral transmission, supply inconsistency). The advent of high-purity recombinant HSA (rHSA) has eliminated these barriers.
Comparison: Plasma vs. Recombinant

Regulatory Precedent: The ERVEBO Case Study
The safety and efficacy of InVitria’s rHSA platform have been validated in the most demanding regulatory contexts.
- ERVEBO (Merck): The FDA and EMA-approved live attenuated Ebola vaccine utilizes recombinant HSA as a critical stabilizer (U.S. Food and Drug Administration [FDA], 2019).
- Implication: This approval sets a regulatory precedent, confirming that rHSA is accepted as a safe, cGMP excipient for injectable biologics, clearing the path for its adoption in mAb formulations to replace polysorbates.
Comparative Analysis: Stability Risk Matrix
The following matrix contrasts the stability profile of a standard Polysorbate 80 formulation against one stabilized with Exbumin® rHSA.
Stability Risk Matrix – Polysorbates vs. rHSA
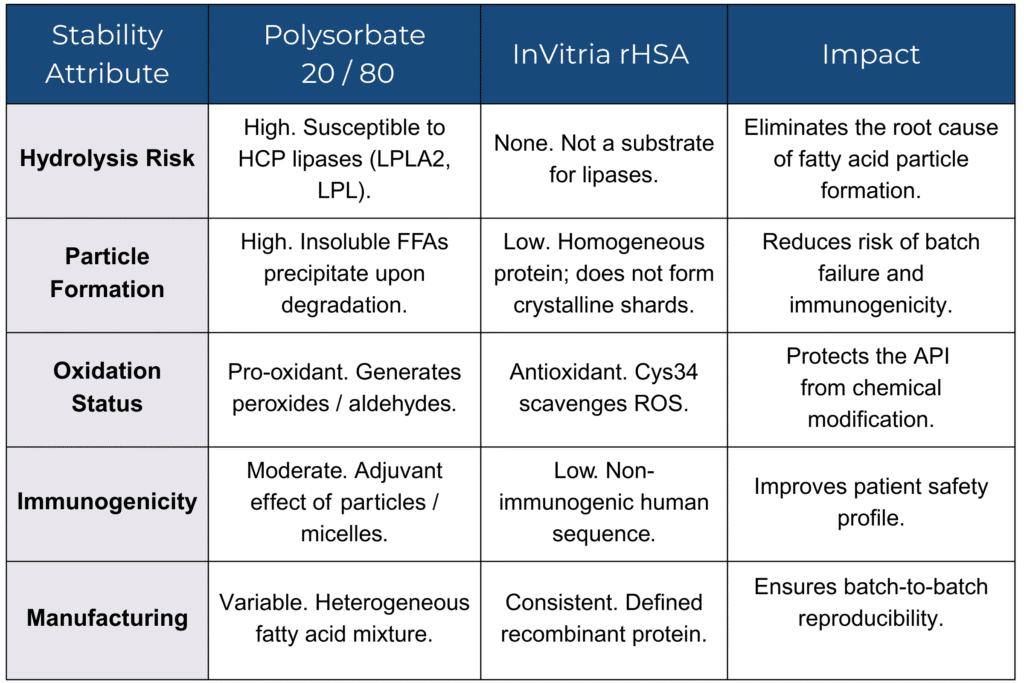
Removing a Known Failure Mode
The biopharmaceutical industry is moving toward higher concentration formulations and more complex modalities, where the liabilities of polysorbates are increasingly untenable. The “necessary evil” is no longer necessary.
The data demonstrates that replacing polysorbates with InVitria’s Exbumin & Optibumin eliminates the risks of enzymatic hydrolysis and fatty acid particle formation while providing active antioxidant protection. By transitioning to a blood-free, regulatory-approved recombinant albumin, formulators can align with modern Quality by Design (QbD) principles by removing a known degradation pathway to ensure the safety, stability, and efficacy of life-saving therapeutics.

The following content is gated. Please, subscribe to open access to it.
Footnotes
References
- Hall, T., Sandefur, S. L., Frye, C. C., Tuley, T. L., & Huang, L. (2016). Polysorbates 20 and 80 degradation by group XV lysosomal phospholipase A2 isomer X1 in monoclonal antibody formulations. Journal of Pharmaceutical Sciences, 105(5), 1633–1642. https://jpharmsci.org/article/S0022-3549(16)00388-9/
- Chiu, J., Valente, K. N., Levy, N. E., Min, L., Lenhoff, A. M., & Lee, K. H. (2017). Knockout of a difficult-to-remove CHO host cell protein, lipoprotein lipase, for improved polysorbate stability in monoclonal antibody formulations. Biotechnology and Bioengineering, 114(5), 1006–1015. https://doi.org/10.1002/bit.26237
- Kishore, R. S. K., Kiese, S., Fischer, S., Pappenberger, A., Grauschopf, U., & Mahler, H. C. (2011). Degradation of polysorbates 20 and 80: Studies on thermal autoxidation and hydrolysis. Journal of Pharmaceutical Sciences, 100(2), 721–731. https://jpharmsci.org/article/S0022-3549(15)32291-7/fulltext
- Saggu, M., Patel, A. R., & Koulov, A. V. (2015). Identification of subvisible particles in biopharmaceutical formulations using Raman spectroscopy provides insight into polysorbate 20 degradation pathway. Pharmaceutical Research, 32(9), 2877–2888. https://doi.org/10.1007/s11095-015-1670-x
- Kerwin, B. A. (2008). Polysorbates 20 and 80 used in the formulation of protein biotherapeutics: Structure and degradation pathways. Journal of Pharmaceutical Sciences, 97(8), 2924–2935. https://doi.org/10.1002/jps.21190
- Wang, W., Vlasak, J., Li, Y., Pristatsky, P., Fang, Y., Pittman, T., Roman, J., Wang, Y., Prueksaritanont, T., & Ionescu, R. (2011). Impact of methionine oxidation in human IgG1 Fc on serum half-life of monoclonal antibodies. Molecular Immunology, 48(6–7), 860–866. https://doi.org/10.1016/j.molimm.2010.12.009
- Singh, S. K. (2011). Impact of product-related factors on immunogenicity of biotherapeutics. Journal of Pharmaceutical Sciences, 100(2), 354–387. https://jpharmsci.org/article/S0022-3549(15)32277-2/fulltext
- Boven, K., Stryker, S., Knight, J., Thomas, A., van Regenmortel, M., Kemeny, D. M., & Casadevall, N. (2005). Pure red cell aplasia and recombinant erythropoietin. New England Journal of Medicine, 352(20), 2021–2032.
https://www.nejm.org/doi/full/10.1056/NEJM200208083470623 - Sønderby, P., Bukrinski, J. T., Hebditch, M., Peters, G. H. J., Curtis, R. A., & Harris, P. (2018). Self-interaction of human serum albumin: A formulation perspective. ACS Omega, 3(11), 16105–16117. https://doi.org/10.1021/acsomega.8b02245
- Khan, S., & Naeem, A. (2024). Serum albumin as a probable extracellular chaperone. Archive of Biochemistry. https://www.chemisgroup.us/articles/AB-7-109.pdf
- Oettl, K., & Stauber, R. E. (2007). Physiological and pathological changes in the redox state of human serum albumin critically influence its binding properties. British Journal of Pharmacology, 151(5), 580–590. https://doi.org/10.1038/sj.bjp.0707251
- Taverna, M., Marie, A.-L., Mira, J.-P., & Guidet, B. (2013). Specific antioxidant properties of human serum albumin. Annals of Intensive Care, 3, Article 4. https://doi.org/10.1186/2110-5820-3-4
- U.S. Food and Drug Administration. (2019). ERVEBO (Ebola Zaire vaccine, live) package insert.
https://www.fda.gov/media/133748/download