- Home
- Thiol by Combat: Enhanced Covalent Conjugation with Recombinant Albumin Compared to Human Plasma-Derived Albumin
Thiol by Combat: Enhanced Covalent Conjugation with Recombinant Albumin Compared to Human Plasma-Derived Albumin
Published on 3 October 2025
Application Note
Author(s): Mark Stathos, PhD, Applications Scientist, Jacob Weber, PhD, Vice President of Process Development, Vladimir Akoyev, PhD, Vice President of Analytical Development, Marcus Curl, Vice President of Product Applications
InVitria, Inc., USA
EXECUTIVE SUMMARY
Covalent conjugation of drugs and diagnostic imaging reagents to albumin is a versatile and widely used strategy in pharmacology. This chemistry improves drug stability in serum, reduces renal clearance, and increases solubility, while also being highly specific. Conjugation is usually accomplished by reacting a pharmacological agent with the free thiol form of albumin’s cysteine at position 34 (Cys34). However, about 40% of donor-derived human serum albumin (HSA) cannot participate in this reaction because the Cys34 is modified and unavailable. Donor-derived HSA also contains many other post-translational modifications, making it heterogeneous in composition.
In contrast, Optibumin® 25 (rHSA), InVitria’s recombinant albumin, is 99% mercapto-albumin (unmodified free thiol at Cys34). This makes it the ideal partner for efficient, consistent conjugation reactions that yield uniform drug products.
Key Highlights
- 99% reduced Cys34 vs ~60% in plasma HSA
- 10x lower lot-to-lot variability
- 3x higher conjugation efficiency
- Linear and predictable conjugation profiles
- Recombinant, cGMP, animal-origin-free
- Ideal partner for covalent drug conjugation
INTRODUCTION
Covalent conjugation to human serum albumin (HSA) is a powerful strategy in drug development and diagnostics, with applications across a wide range of molecules. Notable examples of this approach include the drug aldoxorubicin for chemotherapy (Gong et al 2018), bioresponsive prodrugs (Yang et al. 2018), near-infrared dyes for diagnostic imaging (Zhang et al. 2023), radioactive isotopes for theranostics (Fischer et al. 2022), peptides for HIV treatment (Xie et al. 2010), and nucleic acids for aptamer generation (Dinesen et al., 2022).Given the widespread use of the conjugation strategy, it is not surprising that it confers several performance benefits.
Low molecular weight drugs such as small molecules or siRNAs are often rapidly filtered out of serum via the kidneys but large molecules like albumin (and substances bound to it) are retained in serum to a much greater extent (Hoogenboezem et al., 2024). Furthermore, albumin has a serum half-life of 19 days (Liu et al., 2020)—much longer than most proteins, because it is recycled via the neonatal Fc receptor (Sand et al., 2015). This is the same mechanism responsible for recycling IgG, and covalently linked moieties benefit from the same half-life extension. Albumin also improves solubility, which is critical for many hydrophobic small molecules (Mehtala et al.,2016). Finally, conjugation is highly specific, typically occurring only at the reduced Cys34 (Yu et al., 2022).
However, ~40% of plasma-derived albumin is oxidized (cysteinylated) and cannot participate in conjugation (Oettl et al., 2010). Levels of oxidation also vary significantly from lot to lot. This variability reduces conjugation efficiency and produces heterogeneous products. Free Cys34 (mercapto-albumin) also functions as a potent physiological antioxidant. Moreover, Cys34 is central to albumin’s antioxidant role (Bruschi et al., 2013), and its oxidation is linked to aging (Nakashima et al., 2018).
To address these issues, InVitria developed Optibumin 25, a highly pure and consistent recombinant human serum albumin (rHSA), that is 99% mercapto-albumin (free thiol at Cys34). Optibumin 25, being animal-origin-free, also satisfies FDA and EMA guidance to eliminate donor-derived raw materials in biologics (EMA, 2025). This makes it ideal for APIs that are hydrophobic, rapidly cleared, unstable in serum, or require covalent conjugation for optimal pharmacokinetics. In this application note, Optibumin 25’s improved Cys34 conjugation efficiency is
evaluated against plasma-derived HSA using Alexa Fluor 488 C5 maleimide as a model conjugation partner.
RESULTS AND DISCUSSION
Cys34 Redox Characterization of Optibumin 25 vs Plasma HSA
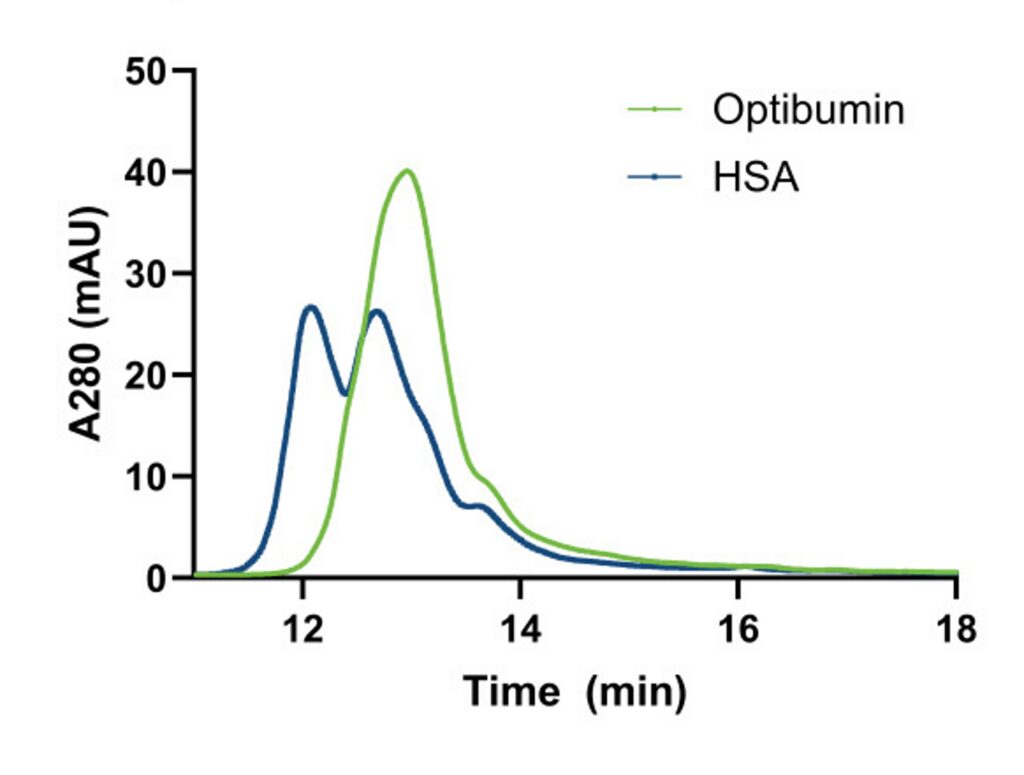
To characterize the free-Cys34 on Optibumin 25 and HSA, high performance liquid chromatography (HPLC) was performed. Representative HPLC chromatograms for Optibumin 25 and HSA used to determine the Cys34 redox status are shown in Figure 1A.
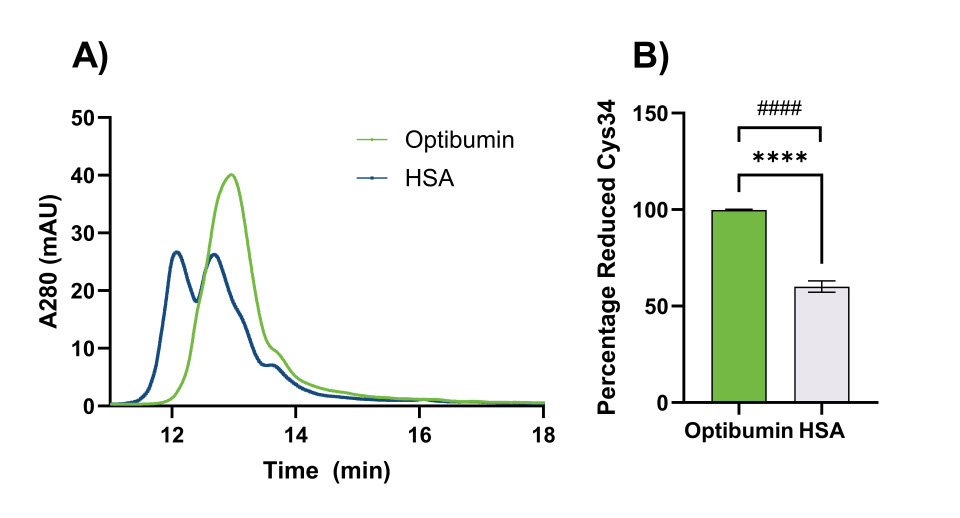
These plots depict the amount of protein present in the sample as it flows through a column and is separated by hydrophobicity over time. Relative protein concentration was measured by absorbance at 280 nm (A280), a measure of the number of aromatic amino acids present. Notably, Optibumin’s chromatogram (green) shows only one peak indicating it is entirely reduced and has minimal additional post translational modifications whereas the overlaid HSA chromatogram (blue) has two main peaks corresponding to oxidized i.e., cysteinylated albumin (left) and reduced albumin (right). Other subpeaks on the HSA chromatogram correspond to post translational modifications on other residues.
To assess the consistency between lots for each albumin, 10 lots of Optibumin 25 and 9 lots of clinical grade HSA were run on HPLC using the same method. The average percentage of free-cysteine for these samples, determined by calculating the area under the curve corresponding to each peak, is shown in Figure 1B. The mean percentage of free thiol (Cys34) for Optibumin 25 lots was 99% whereas the mean for the clinical HSA group was only 60% which was a significant difference (Welch’s t-test, p < 0.0001). Additionally, the standard deviation of reduced cysteine percentage was 0.295% for the Optibumin lots and 2.93% for the HSA lots indicating a nearly 10x higher variation in donor-derived HSA compared to the remarkably consistent cGMP recombinant albumin (Optibumin), which was also a statistically significant difference (F-test of variances, p < 0.0001). Taken together, these results demonstrate that Optibumin is significantly more reduced than plasma HSA. It also has ten times less variation in reduced Cys34 from lot to lot than HSA, and is overall a more uniform product.
These results demonstrate that Optibumin provides a highly uniform and reduced substrate, critical for reproducible conjugation workflows.
Conjugation Efficiency off Optibumin vs Plasma HSA
To evaluate conjugation efficiency, Alexa Fluor 488 C5 maleimide was added to either Optibumin or HSA to achieve a twofold serial dilution of dye to albumin molar ratios from 1.25:1 down to 0.04:1 and a final albumin concentration of 2 mg/mL and incubated for 30 minutes at room temperature in the dark. Additional precautions were taken to prevent nonspecific reaction to lysine residues and hydrolysis of the maleimide moiety (see materials and methods).
HPLC was then performed on the reaction mixtures as described in the initial albumin characterization with the addition of monitoring absorbance at 498 nm (A498) to detect the presence of the AF488Mal dye. Coincidence of the A280 peak (solid lines) and the A498 peak (dashed lines) in Figure 2A indicates that the conjugation reaction was successful. Notably, the left peak on the HSA A280 curve (solid blue) corresponding to cysteinylated Cys34 remains prominent despite the presence of the conjugate dye showing that this species is indeed nonreactive. Additionally, the conjugated HSA curve (dashed blue) is asymmetrical and dissimilar to the total protein HSA curve (solid blue) showing that plasma HSA reacts with the dye in a less predictable manner. In contrast, the shape of the dye conjugated Optibumin curve (dashed green) is similar to the total protein Optibumin curve (solid green) and both curves are highly symmetrical demonstrating that Optibumin reacts uniformly and predictably with the conjugate dye.
To compare the relative dye conjugation efficiency of Optibumin and HSA, the area under the curve (AUC) of each A280 and A498 chromatogram was calculated. These are measures of total protein and total conjugated dye respectively. The ratio of the A498 AUC divided by the A280 AUC is therefore a relative (but not absolute) measure of reaction efficiency that can be compared between samples. This ratio was plotted against the dye-to-protein molar ratios used in the reactions in Figure 2B and the data fitted to a linear regression.
The fit for Optibumin was incredibly precise and highly linear throughout the range of molar ratios tested with an R2 of 0.992. For HSA, the fit was still good with an R2 of 0.9053 but there is noticeable downward curve at higher molar ratios. This nonlinearity indicates that HSA becomes saturated at a molar ratio of 0.16:1 dye to albumin and further increases in conjugation are likely driven by nonspecific interactions to other residues.
The slope of the Optibumin 25 fit was significantly steeper (Fisher’s F-test, p < 0.0001) than the slope of the HSA fit indicating that significantly more labeled albumin is produced per mole of dye added for Optibumin 25 than for HSA. The extent of this difference can be quantified by dividing the slope of the Optibumin 25 line by the slope of the HSA line to get 3.00.
This means that the Cys34 conjugation reaction is three times as efficient with Optibumin than with HSA. Interestingly this is a greater improvement than would be expected based on the difference in available Cys34 shown in Figure 2B. This disparity may be due to post translational modifications and structural heterogeneity compared to Optibumin or increased aggregation compared to Optibumin. Furthermore, the difference in labeling becomes more substantial with dye to protein ratios that approach 1:1 which is the expected stoichiometry of the reaction and thus the condition that is the most realistic use case for this application. Taken together, the highly linear increase in conjugation efficiency up to a 1.25:1 dye-to-protein ratio means that Optibumin is essentially a perfect conjugation partner for maleimide conjugation and demonstrating a threefold higher efficiency and more predictable performance compared to plasma HSA.

CONCLUSION
Optibumin 25, InVitria’s animal-origin-free recombinant albumin, outperformed plasma-derived albumin in a model covalent conjugation application in terms of both efficiency and consistency. This advantage is due to Optibumin 25’s high mercapto-albumin (free Cys34) content, which enables nearly every molecule to efficiently participate in the conjugation reaction. In contrast, plasma-derived albumin is uncontrolled, with ~40% of Cys34 unavailable because it is cysteinylated. In practice, this translated to a threefold improvement in conjugation efficiency with Optibumin 25 compared to donor-derived HSA, as measured by the relative amount of conjugated dye per protein determined by HPLC. The difference in reduced Cys34 content alone does not fully explain this disparity, suggesting that additional post-translational modifications and structural heterogeneity of plasma-derived albumin also impair conjugation. Together, these results establish Optibumin 25 as the analytically superior and regulatory-aligned choice for covalent conjugation applications.
MATERIALS & METHODS
Cys34- Maleimide Conjugation Reaction
Alexa Fluor™ 488 C5 Maleimide (AF488Mal,Thermo-Fisher Scientific) was reconstituted in DMSO according to the manufacturer’s instructions and subsequently diluted in PBS that had been previously degassed and adjusted to pH 6. Degassing is important for minimizing hydrolysis of the maleimide which renders it unable to react and reducing the pH is important for minimizing unwanted cross-reactivity of the maleimide with primary amines found on lysine residues in albumin (Kirkpatrick et al. 2024).
Notably, this dye contains a short five carbon linker that helps the maleimide reach the Cys34 which is found in a small pocket on the surface of albumin (Kratz et al 2002). It is possible that the reaction can be hampered by steric hindrance if a linker is not
employed.
HPLC
Reverse-phase HPLC (RP-HPLC) was conducted on a 1290 Infinity II HPLC System (Agilent Technologies) with Quaternary Pump (G7104A) and 1290 Infinity II DAD FS Detector (G7117A) and an Agilent InfinityLab® Bio-Inert Max-Light® Cartridge Cell, 10 mm (G5615-60018). Chromatographic separation was achieved using an ACQUITY ™ UPLC Protein BEH C4 Column (300 Å pore size, 1.7 µm particle size, 2.1 × 150 mm; 186004497, Waters Corporation).
Mobile Phase A consisted of LC-MS grade water with 0.1% trifluoroacetic acid (TFA), and Mobile Phase B consisted of 100% acetonitrile with 0.1% TFA. A shallow gradient of Mobile Phase B (35% to 40%) was applied to resolve two major albumin species. Detection of albumin species was performed at 215 nm and 280 nm, while free Alexa Fluor 488 C5 Maleimide dye and dye-conjugated albumin were monitored at 498 nm. Data acquisition and analysis were conducted using Agilent MassHunter® Software (Agilent Technologies).
Applications & Use Cases
Optibumin 25 supports reliable conjugation in a wide range of drug and diagnostic workflows, including:
- Diagnostic imaging reagents – ensure consistent conjugation of dyes and tracers
- Theranostics and radiolabeled drugs – enable efficient binding to albumin for extended circulation
- Hydrophobic APIs – improve solubility and pharmacokinetics through covalent albumin binding
Regulatory Relevance
- Animal-origin-free (AOF): eliminates plasma-derived variability and risk
- cGMP-manufactured: designed for clinical and commercial use
- Regulatory alignment: meets FDA and EMA guidance for eliminating donor-derived raw materials in biologics
Featured Solution
Optibumin 25 – Recombinant Human Serum Albumin for Conjugation Workflows
Optibumin 25 is a recombinant, animal-origin-free human serum albumin with 99% free Cys34, providing a highly consistent substrate for covalent drug conjugation. Unlike plasma-derived HSA, it delivers 3x higher conjugation efficiency and 10x lower variability, supporting reproducible, scalable, and regulatory-compliant biomanufacturing.
Frequently Asked Questions (FAQs)
Q: Why is Cys34 important for drug conjugation?
Cys34 is the only free thiol in albumin and is the primary site for covalent conjugation. Having a high percentage of free Cys34 enables efficient, predictable attachment of conjugates.
Q: How does Optibumin 25 differ from plasma-derived HSA?
Optibumin 25 is 99% free Cys34, compared to ~60% in plasma HSA. It also shows 10x lower lot-to-lot variability and delivers 3x higher conjugation efficiency.
Q: What are the benefits of using recombinant albumin for conjugation workflows?
Recombinant albumin improves consistency, reduces variability, eliminates donor-derived risks, supports regulatory compliance, and enhances conjugation efficiency.
See the complete analysis comparing recombinant and plasma-derived albumin in covalent drug conjugation.
The following content is gated. Please, subscribe to open access to it.
Footnotes
References
- Bruschi, M., Candiano, G., Santucci, L., & Ghiggeri, G. M. (2013). Oxidized albumin: The long way of a protein of uncertain function. Biochimica et Biophysica Acta (BBA) – General Subjects, 1830(12), 5473–5479. https://doi.org/10.1016/j.bbagen.2013.04.017
- Dinesen, A., Winther, A., Wall, A., Märcher, A., Palmfeldt, J., Chudasama, V., Wengel, J., Gothelf, K. V., Baker, J. R., & Howard, K. A. (2022). Albumin biomolecular drug designs stabilized through improved thiol conjugation and a modular locked nucleic acid functionalized assembly. Bioconjugate Chemistry, 33(2), 333–342. https://doi.org/10.1021/acs.bioconjchem.1c00561
- European Medicines Agency. (2025). Guideline on quality, non-clinical and clinical requirements for investigational advanced therapy medicinal products in clinical trials (EMA/CAT/22473/2025). https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-quality-non-clinical-clinical-requirements-investigational-advanced-therapy-medicinal-products-clinical-trials_en.pdf
- Fischer, N. H., Lopes van den Broek, S. I., Herth, M. M., & Diness, F. (2022). Radiolabeled albumin through SNAr of cysteines as a potential pretargeting theranostic agent. RSC Advances, 12, 35032–35036. https://doi.org/10.1039/D2RA06406E
- Gong, J., Yan, J., Forscher, C., & Hendifar, A. (2018). Aldoxorubicin: A tumor-targeted doxorubicin conjugate for relapsed or refractory soft tissue sarcomas. Drug Design, Development and Therapy, 12, 777–786. https://doi.org/10.2147/DDDT.S140638
- Hoogenboezem, E. N., Patel, S. S., Lo, J. H., Cavnar, A. B., Babb, L. M., Francini, N., Gbur, E. F., Patil, P., Colazo, J. M., Michell, D. L., Sanchez, V. M., McCune, J. T., Ma, J., DeJulius, C. R., Lee, L. H., Rosch, J. C., Allen, R. M., Stokes, L. D., Hill, J. L., … Duvall, C. L. (2024). Structural optimization of siRNA conjugates for albumin binding achieves effective MCL1-directed cancer therapy. Nature Communications, 15(1), Article 1581. https://doi.org/10.1038/s41467-024-45609-0
- Kirkpatrick, B. E., Anseth, K. S., & Hebner, T. S. (2024). Diverse reactivity of maleimides in polymer science and beyond. Polymer International. Advance online publication. https://doi.org/10.1002/pi.6715
- Kratz, F., Warnecke, A., Scheuermann, K., Stockmar, C., Schwab, J., Lazar, P., Drückes, P., Esser, N., Drevs, J., Rognan, D., Bissantz, C., Hinderling, C., Folkers, G., Fichtner, I., & Unger, C. (2002). Probing the cysteine-34 position of endogenous serum albumin with thiol-binding doxorubicin derivatives: Improved efficacy of an acid-sensitive doxorubicin derivative with specific albumin-binding properties compared to that of the parent compound. Journal of Medicinal Chemistry, 45(25), 5523–5533. https://doi.org/10.1021/jm0204946
- Liu, X., Mohanty, R. P., Maier, E. Y., Peng, X., Wulfe, S., Looney, A. P., Aung, K. L., & Ghosh, D. (2020). Controlled loading of albumin-drug conjugates ex vivo for enhanced drug delivery and antitumor efficacy. Journal of Controlled Release, 328, 1–12. https://doi.org/10.1016/j.jconrel.2020.08.015
- Mehtala, J. G., Kulczar, C., Lavan, M., Knipp, G., & Wei, A. (2015). Cys34-PEGylated human serum albumin for drug binding and delivery. Bioconjugate Chemistry, 26(5), 941–949. https://doi.org/10.1021/acs.bioconjchem.5b00143
- Nakashima, F., Shibata, T., Kamiya, K., Yoshitake, J., Kikuchi, R., Matsushita, T., Ishii, I., Giménez-Bastida, J. A., Schneider, C., & Uchida, K. (2018). Structural and functional insights into S-thiolation of human serum albumins. Scientific Reports, 8, Article 932. https://doi.org/10.1038/s41598-017-18860-9
- Oettl, K., & Marsche, G. (2010). Redox state of human serum albumin in terms of cysteine-34 in health and disease. In Methods in Enzymology (Vol. 474, pp. 181–195). https://doi.org/10.1016/S0076-6879(10)74011-8
- Sand, K. M. K., Bern, M., Nilsen, J., Noordzij, H. T., Sandlie, I., & Andersen, J. T. (2015). Unraveling the interaction between FcRn and albumin: Opportunities for design of albumin-based therapeutics. Frontiers in Immunology, 5, Article 682. https://doi.org/10.3389/fimmu.2014.00682
- Xie, D., Yao, C., Wang, L., Min, W., Xu, J., Xiao, J., Huang, M., Chen, B., Liu, B., Li, X., & Jiang, H. (2010). An albumin- conjugated peptide exhibits potent anti-HIV activity and long in vivo half-life. Antimicrobial Agents and Chemotherapy, 54(1), 191–196. https://doi.org/10.1128/AAC.00667-10
- Yang, J., Lv, Q., Wei, W., Yang, Z., Dong, J., Zhang, R., Kan, Q., He, Z., & Xu, Y. (2018). Bioresponsive albumin-conjugated paclitaxel prodrugs for cancer therapy. Drug Delivery, 25(1), 807–814. https://doi.org/10.1080/10717544.2018.1451935
- Yu, L., Hua, Z., Luo, X., Zhao, T., & Liu, Y. (2022). Systematic interaction of plasma albumin with the efficacy of chemotherapeutic drugs. Biochimica et Biophysica Acta (BBA) – Reviews on Cancer, 1877(1), Article 188655.
https://doi.org/10.1016/j.bbcan.2021.188655 - Zhang, Y., Jia, Y., & Zhu, S. (2023). NIR-II cyanine@albumin fluorophore for deep tissue imaging and imaging-guided surgery. SmartMat,4(1). https://doi.org/10.1002/smm2.1245
