- Home
- Superior Cryopreservation of T Cells in Low-DMSO, Animal-Origin-Free Formulation Using Optibumin 25 and PentaHibe Base
Superior Cryopreservation of T Cells in Low-DMSO, Animal-Origin-Free Formulation Using Optibumin 25 and PentaHibe Base
Published on 21 August 2025
Application Note
Authors: Andrew Hamann, PhD and Mark Stathos, PhD, InVitria, Inc., USA
In collaboration with PentaHibe, developed by Pharmacosmos A/S, Denmark
EXECUTIVE SUMMARY
This application note presents a defined, animal-origin-free (AOF) cryopreservation strategy addressing the limitations of high-DMSO and serum-derived components. The formulation—10% PentaHibe Base, 2% DMSO, and 4% Optibumin 25—was tested in CAR-T–mimicking workflows using healthy donor T cells.
PentaHibe Base is a non-penetrating, carbohydrate-based cryoprotectant derived from pentaisomaltose, a patented subfraction of Dextran 1 with a well-established clinical safety profile. Optibumin 25 is a recombinant human serum albumin (HSA) produced under cGMP in a non-mammalian system.
The formulation was benchmarked against plasma-derived HSA and CryoStor CS10, CS5, and CS2. Despite using significantly less DMSO than CS10 and CS5—and the same concentration as CS2—it matched or exceeded post-thaw viability and proliferation across all donors and endpoints. These results support the use of this fully defined, animal-origin-free formulation in GMP-aligned cryopreservation workflows for adoptive cell therapies, including CAR-T.
Key Highlights
- Maintains high post-thaw viability with only 2% DMSO
- Matches or exceeds CS10 and plasma-derived HSA
- Outperforms CS2 across all donors and endpoints
- Supports strong T cell proliferation at 72 hours
- Defined, animal-origin-free composition suitable for GMP workflows
INTRODUCTION
Cryopreservation is a critical step in the manufacturing and delivery of T cell–based therapies, directly impacting post-thaw viability, proliferation, and overall therapeutic efficacy. Conventional protocols often rely on high concentrations of dimethyl sulfoxide (DMSO) and plasma-derived human serum albumin (HSA), which, while effective, present well-documented risks. DMSO is associated with cytotoxicity and infusion–related side effects (Madsen et al., 2018), while blood-derived HSA introduces concerns related to donor variability and the potential for pathogen transmission (MacLennan & Barbara, 2006; Lu et al., 2024).
To improve safety, consistency, and regulatory compliance, agencies such as the U.S. FDA have issued guidance encouraging the use of chemically defined, animal-origin-free (AOF) materials in advanced therapy manufacturing (U.S. FDA, 2024). This shift is especially critical for CAR-T and adoptive cell therapies, where product reproducibility and patient safety are paramount.
PentaHibe® Base (Pharmacosmos), offers a novel approach to cryopreservation. It is a non-penetrating, carbohydrate-based cryoprotectant. It is derived from pentaisomaltose, a patented subfraction of Dextran 1 with a well-established clinical safety profile. Unlike traditional intracellular cryoprotectants, PentaHibe acts extracellularly, avoiding membrane penetration and minimizing intracellular toxicity. Its clinical safety is supported by more than eight million doses of its parent compound (Ljungström, 2006), and it has demonstrated strong performance at reduced DMSO concentrations (2%), maintaining membrane integrity and intracellular stability. Previous studies have shown that PentaHibe, when used with plasma-derived albumin, supports post-thaw T cell viability and functionality—matching conventional 10% DMSO formulations (Haastrup et al., 2021). Similar success has been observed in other cell types, such as MSCs (Svalgaard et al., 2020).
Optibumin® 25 (InVitria) is a recombinant human serum albumin (rHSA) produced under cGMP in a non-mammalian expression system. It eliminates the risks associated with plasma-derived HSA while providing equivalent or superior support for cell viability, oxidative protection, and osmotic balance.
Internal studies have shown that Optibumin 25 improves post-thaw recovery and proliferation of T cells compared to clinical-grade HSA (InVitria, 2024), while also enabling reduced DMSO usage in other cell types, including MSCs and iPSCs (InVitria, 2025). For additional data on Optibumin® 25 in cryopreservation, see InVitria’s application notes on:
- Enhanced cryopreservation of T cells using Optibumin rHSA
- Low-DMSO cryopreservation of MSCs
- Low-DMSO cryopreservation of iPSCs
In this study, we evaluated a fully animal-origin-free (AOF) cryopreservation formulation composed of 10% (v/v) PentaHibe® Base, 2% (v/v) DMSO, and 4% (v/v) Optibumin 25. Its performance was compared to PentaHibe Base + blood-derived HSA (using the same ratios), and to commercial CryoStor formulations CS10, CS5, and CS2. To reflect clinically relevant CAR-T manufacturing workflows, T cells were expanded for six days, cryopreserved at 10 × 10⁶ cells/mL, and held in cryopreservation media for one hour post-thaw prior to plating (Li, 2019). Post-thaw viability (0 h and 24 h) and proliferation (72 h) were assessed across three donors to evaluate the formulation’s suitability for clinical T cell manufacturing.
RESULTS AND DISCUSSION
Comparable T Cell Cryopreservation with Optibumin 25 and Plasma-Derived HSA
T cells cryopreserved in the PentaHibe® Base + Optibumin 25 formulation exhibited strong post-thaw viability and proliferation across all three donors, performing comparably to the PentaHibe Base + HSA control (Figure 1). Immediately after thawing (T0), viabilities ranged from 75–83% for Optibumin 25 and 75–86% for HSA. As expected following cryopreservation, viability declined by 24 hours (T24); however, the Optibumin 25 formulation preserved viability within 44–59%, closely matching the HSA group (41–59%) and demonstrating reduced viability loss during the extended recovery window.
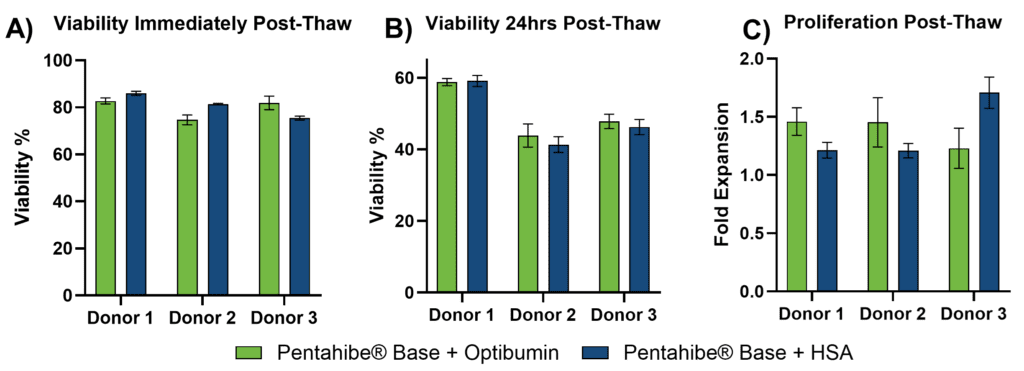
T cells from three donors were cryopreserved using PentaHibe Base + 2% DMSO with either 4% Optibumin 25 (recombinant HSA) or 4% plasma-derived HSA. Post-thaw outcomes include (A) viability at thaw (T0), (B) viability at 24 hours (T24), and (C) fold expansion at 72 hours. Bars represent the mean ± SD of triplicate wells. Optibumin 25 performed comparably to plasma-derived HSA across all endpoints in a low-DMSO setting.
Optibumin 25 with PentaHibe Base Matches or Surpasses CryoStor Performance With Significantly Less DMSO
When compared to commercial CryoStor formulations, the PentaHibe Base + Optibumin 25 solution consistently matched or outperformed CryoStor CS10 and CS5, despite containing only 2% DMSO (Figure 2). At T0, viability was comparable across donors (75–83%), meeting or exceeding values from CS10 and CS5 and notably higher than CS2 in Donor 2.
At T24, viability in the Optibumin 25 group (44–59%) remained comparable to CS10 and CS5 and was significantly higher than CS2 across all donors (p < 0.001), highlighting the formulation’s protective effect during delayed post-thaw stress. These data are consistent with earlier internal and published findings showing the benefit of albumin-based cryoprotection during extended recovery windows.
Proliferation data at 72 hours post-thaw further supported the formulation’s effectiveness. Optibumin 25-treated cells expanded 1.23–1.46-fold, closely matching CS10 (1.48–1.49) and outperforming CS2 (1.15–1.25), with a significant difference observed in Donor 2 (p < 0.001). These results demonstrate that the PentaHibe Base + Optibumin 25 formulation provides robust cryoprotection and supports post-thaw performance equivalent to or better than commercial cryopreservation media containing substantially higher DMSO levels.
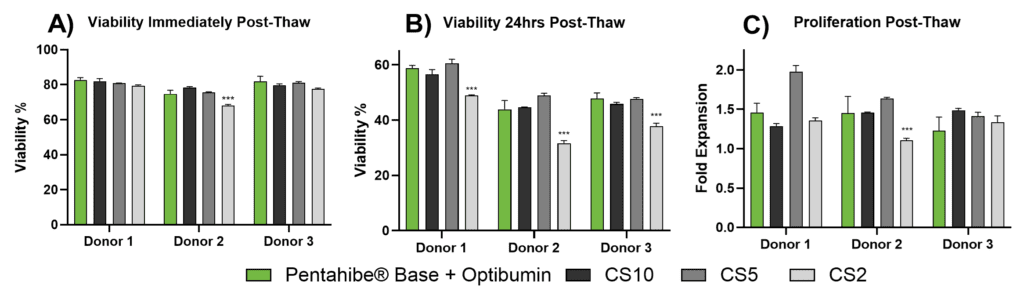
T cell cryopreservation outcomes with PentaHibe Base + Optibumin 25 vs CryoStor formulations (CS10, CS5, CS2). Assessed metrics: (A) viability at T0 (thaw), (B) viability at T24 (24 hours post-thaw), and (C) fold expansion at 72 hours. PentaHibe Base + Optibumin 25 matched or exceeded CS10/CS5 and significantly outperformed CS2 (p < 0.001, one-way ANOVA).
In summary, PentaHibe Base + Optibumin 25 generally matched or exceeded the performance CryoStor CS10 and CS5, despite using 2–5x less DMSO. Notably, the PentaHibe Base + Optibumin 25 formulation outperformed CryoStor CS2 despite both containing the same 2% DMSO, highlighting the added benefit of combining a non-penetrating carbohydrate cryoprotectant with recombinant albumin. These results demonstrate that PentaHibe Base + Optibumin effectively supports post-thaw recovery and expansion in a defined, nimal-origin-free system compatible with clinical manufacturing needs. These findings build on previous studies using PentaHibe Base + HSA, which demonstrated comparable or superior T cell recovery and function relative to conventional 10% DMSO systems (Haastrup et al., 2021). Our results extend this evidence by demonstrating that Optibumin 25 can successfully replace blood-derived albumin, enabling a fully AOF cryopreservation platform that minimizes DMSO exposure without compromising post-thaw cell quality.
CONCLUSION
This study demonstrates that a fully animal-origin-free cryopreservation formulation—10% PentaHibe Base, 2% DMSO, and 4% Optibumin 25—effectively preserves T cell viability and supports robust proliferation post-thaw. Across three donors, it performed comparably to PentaHibe + plasma-derived HSA and matched or exceeded CryoStor CS10 and CS5, despite significantly lower DMSO content.
These findings are consistent with Haastrup et al. (2021), who reported strong outcomes using PentaHibe + HSA under ideal lab conditions, including fresh donor cells, a higher freezing density (20 × 10⁶ cells/mL), and a controlled-rate freezer. In contrast, the present study used previously expanded and frozen T cells, a lower freezing density (10 × 10⁶ cells/mL), and a passive freezing method with a 1-hour post-thaw hold—design choices intended to better simulate real-world CAR-T manufacturing conditions. This strengthens the relevance of our findings for clinical workflows.
Together, the results validate this defined, low-toxicity formulation as a safe and scalable solution for T cell cryopreservation. It eliminates reliance on animal- and serum-derived components, reduces DMSO burden, and supports GMP-compliant manufacturing in adoptive cell therapy processes.
MATERIALS AND METHODS
T Cell Source and Expansion
Human peripheral blood T cells were isolated from leukopaks (three donors) and cryopreserved immediately in CryoStor CS10 (BioLife Solutions) for subsequent experiments. To mimic CAR-T cell manufacturing workflows, cells were thawed, activated with anti-CD3/CD28 stimulation for 3 days, and subsequently expanded in complete growth medium for an additional 3 days.
Cryopreservation
Cryopreservation media were prepared fresh on the day of use (Pharmacosmos A/S, 2024). The experimental AOF formulation contained 10% PentaHibe Base (Pharmacosmos), 2% DMSO (Sigma-Aldrich), and 4% Optibumin 25 (InVitria). Comparator conditions included:
- PentaHibe Base + HSA (10% PentaHibe Base, 2% DMSO, and 4% blood-derived HSA)
- CryoStor CS10, CS5, and CS2 (BioLife Solutions) containing 10%, 5%, and 2% DMSO, respectively
T cells were resuspended at 10 × 10⁶ cells/mL in each formulation and aliquoted into cryovials (1 mL per vial). Cryovials were frozen using a passive-rate freezing device (Mr. Frosty™, Thermo Fisher Scientific) in a −80°C freezer overnight before transfer to vapor-phase liquid nitrogen for long-term storage. For thawing, vials were rapidly warmed in a 37°C water bath. To mimic CAR-T clinical workflows, thawed cells were held in the cryopreservation formulations at room temperature for 1 hour before being plated for analysis (Li, 2019).
Viability and Expansion Analysis
Post-thaw viability was measured immediately (0 h), and again at 24 hours using live-cell imaging with viability dye over 72 hours. Fold expansion was calculated by normalizing viable cell counts to initial values at time zero.
Featured Solutions
Optibumin 25 – Recombinant Human Albumin for Low-DMSO Cryopreservation
 Optibumin 25 is a recombinant, animal-origin-free human serum albumin (rHSA) that replaces plasma-derived HSA in cryopreservation workflows. In this study, Optibumin 25 supported high post-thaw T cell viability and proliferation at only 2% DMSO, performing comparably to CryoStor CS10 and CS5 and significantly better than CS2. By eliminating variability and safety risks linked to blood-derived albumin, Optibumin 25 provides a defined, consistent, and regulatory-friendly solution for T cell and CAR-T manufacturing
Optibumin 25 is a recombinant, animal-origin-free human serum albumin (rHSA) that replaces plasma-derived HSA in cryopreservation workflows. In this study, Optibumin 25 supported high post-thaw T cell viability and proliferation at only 2% DMSO, performing comparably to CryoStor CS10 and CS5 and significantly better than CS2. By eliminating variability and safety risks linked to blood-derived albumin, Optibumin 25 provides a defined, consistent, and regulatory-friendly solution for T cell and CAR-T manufacturing
PentaHibe® Base – Carbohydrate-Based Cryoprotectant
 PentaHibe Base is a carbohydrate-based cryoprotectant developed by Pharmacosmos A/S. Derived from pentaisomaltose, a patented Dextran 1 subfraction, PentaHibe acts extracellularly to protect cells during freeze–thaw cycles while minimizing intracellular toxicity. In this study, PentaHibe Base combined with Optibumin 25 enabled robust T cell viability and proliferation with only 2% DMSO, supporting safer and more scalable workflows for adoptive cell therapies
PentaHibe Base is a carbohydrate-based cryoprotectant developed by Pharmacosmos A/S. Derived from pentaisomaltose, a patented Dextran 1 subfraction, PentaHibe acts extracellularly to protect cells during freeze–thaw cycles while minimizing intracellular toxicity. In this study, PentaHibe Base combined with Optibumin 25 enabled robust T cell viability and proliferation with only 2% DMSO, supporting safer and more scalable workflows for adoptive cell therapies
Learn more about PentaHibe Base
Frequently Asked Questions (FAQs)
Q: Why is low-DMSO important in T cell cryopreservation?
A: High concentrations of DMSO are cytotoxic and can cause infusion-related side effects. Using just 2% DMSO with Optibumin 25 and PentaHibe Base maintains high T cell viability while reducing toxicity.
Q: How does Optibumin 25 improve cryopreservation compared to plasma-derived HSA?
A: Optibumin 25 is recombinant human serum albumin produced without animal or human plasma. It eliminates donor variability and pathogen risk while supporting equal or better post-thaw recovery of T cells.
Q: What makes this cryopreservation method animal-origin-free?
A: The formulation replaces plasma-derived HSA with Optibumin 25 and pairs it with PentaHibe Base, a carbohydrate cryoprotectant. Both are produced under GMP conditions without animal inputs, supporting regulatory compliance.
Q: Is this method compatible with CAR-T cell therapy workflows?
A: Yes. The formulation was tested in CAR-T–mimicking workflows using healthy donor T cells and demonstrated strong post-thaw viability and proliferation at clinically relevant conditions.
Q: Does reducing DMSO affect T cell proliferation after thawing?
A: No. Even at 2% DMSO, T cells cryopreserved with Optibumin 25 and PentaHibe Base matched or exceeded proliferation seen with 10% DMSO CryoStor formulations.
Download the Full Application Note

The following content is gated. Please, subscribe to open access to it.
Footnotes
References
- Burnham, R. E., et al. (2021). Human serum albumin and chromatin condensation rescue ex vivo expanded γδ T cells from the effects of cryopreservation. Cryobiology, 99, 78–87. https://doi.org/10.1016/j.cryobiol.2021.01.011
- Haastrup, E. K., et al. (2021). DMSO (Me2SO) concentrations of 1–2% in combination with pentaisomaltose are effective for cryopreservation of T cells. Transfusion and Apheresis Science, 60(4), 103138. https://doi.org/10.1016/j.transci.2021.103138
- InVitria. (2025). Low-DMSO cryopreservation of iPSCs with Optibumin® 25 recombinant albumin [Application Note]. https://invitria.com/resources/low-dmso-cryopreservation-of-ipscs-with-optibumin-25/
- InVitria. (2024). Enhanced cryopreservation of T cells using Optibumin® rHSA [Application Note]. https://invitria.com/resources/enhanced-cryopreservation-of-t-cells-using-optibumin-rhsa/
- InVitria. (2025). Low-DMSO cryopreservation of MSCs with Optibumin® 25 recombinant albumin [Application Note]. https://invitria.com/resources/low-dmso-cryopreservation-of-mscs-with-optibumin-25-recombinant-albumin/
- Li, R., et al. (2019). Preservation of cell-based immunotherapies for clinical trials. Cytotherapy, 21(9), 943–957. https://doi.org/10.1016/j.jcyt.2019.07.004
- Lu, H., Zhang, Y., & Liu, P. (2024). Identifying new safety risk of human serum albumin: A retrospective study of real-world data. Frontiers in Pharmacology, 15, 1319900. https://doi.org/10.3389/fphar.2024.1319900
- Ljungström, K. G. (2006). Invited commentary: Pretreatment with dextran 1 makes dextran 40 therapy safer. Journal of Vascular Surgery, 43(5), 1070–1072. https://doi.org/10.1016/j.jvs.2005.11.056
- MacLennan, S., & Barbara, J. A. J. (2006). Risks and side effects of therapy with plasma and plasma fractions. Best Practice & Research Clinical Haematology, 19(1), 169–189. https://doi.org/10.1016/j.beha.2005.01.033
- Madsen, B. K., et al. (2018). Adverse reactions of dimethyl sulfoxide in humans: A systematic review. F1000Research, 7. https://doi.org/10.12688/f1000research.16642.2
- U.S. Food and Drug Administration. (2024). Considerations for the use of human- and animal-derived materials in the manufacture of cell and gene therapy and tissue-engineered medical products. FDA-2024-D-1244. https://www.regulations.gov/docket/FDA-2024-D-1244
- Pharmacosmos A/S. (2024). DMSO-reduced cryopreservation using 10% PentaHibe® Base, 2% DMSO and 4% human albumin. https://pentahibe.com/dataAndDocuments
- Pi, C.-H., et al. (2020). Understanding the freezing responses of T cells and other subsets of human peripheral blood mononuclear cells using DSMO-free cryoprotectants. Cytotherapy, 22(5), 291–300. https://doi.org/10.1016/j.jcyt.2020.01.013