- Home
- Improved iPSC Cryopreservation with Optibumin 25: Enhanced Viability, Expansion, and Pluripotency
Improved iPSC Cryopreservation with Optibumin 25: Enhanced Viability, Expansion, and Pluripotency
Published on 25 August 2025
Application Note
Authors: Mark Stathos, PhD, Product Applications Scientist & Andrew Hamann, PhD, Product Applications Scientist
InVitria, Inc., USA
Audio Summary: Improved iPSC Cryopreservation with Optibumin 25
EXECUTIVE SUMMARY
Optibumin 25, InVitria’s recombinant animal-origin-free human serum albumin, improves post-thaw viability, expansion, and maintenance of pluripotency in iPSCs when used in cryopreservation formulations. Compared to plasma-derived HSA, Optibumin 25 demonstrated
significantly greater improvements in cell health and survival. Additionally, supplementing CryoStor® CS5 with 5% Optibumin 25 outperformed CryoStor CS10 alone, enabling a reduction in DMSO concentration from 10% to 5%.
In this application note, we demonstrate how Optibumin 25 enhances iPSC cryopreservation outcomes while supporting serum-free, chemically defined, and animal-origin-free workflows.
Key Highlights
- Improves iPSC post-thaw viability and expansion
- Enables DMSO reduction without compromising results
- Maintains high levels of pluripotency markers
- Outperforms plasma-derived HSA across key metrics
- Animal-origin-free and GMP-grade recombinant albumin
INTRODUCTION
Induced pluripotent stem cells (iPSCs) have enormous potential in clinical regenerative medicine. Derived from reprogrammed adult tissue, these cells can differentiate into nearly any other cell type (Takahashi and Yamanaka, 2006). Clinically relevant examples include dopaminergic neurons for Parkinson’s disease (Sawamoto et al., 2025), beta islet cells for type 1 diabetes (Reichman et al., 2025), cardiomyocytes for heart failure (Zhang et al., 2025), and T cells for CAR-T therapies (Chang et al., 2019).
However, iPSCs are sensitive and difficult to expand consistently while maintaining phenotype, making them costly to work with (Dobruskin et al., 2024). Cryopreservation adds further stress, exposing cells to osmotic and oxidative damage, as well as cytotoxic cryoprotectants. Post-thaw expansion is often slow, especially without optimized protocols (Uhrig et al., 2022). Dimethyl sulfoxide (DMSO), the most widely used cryoprotective agent, has been shown to negatively impact iPSC pluripotency (Sambo et al., 2019) and is cytotoxic.
Albumin is commonly used to mitigate osmotic and oxidative stress, but leading iPSC researchers have historically avoided it. For example, James Thomson’s group developed Essential 8 (E8) media to eliminate albumin, citing batch variability from both animal and human sources as a source of inconsistency in ES and iPS cell culture (Chen et al., 2019).
Optibumin 25, being recombinant and animal-orgin- free, avoids these concerns. It is GMP-compliant and available in closed-system formats for clinical use. We previously demonstrated that Optibumin 25 improves cryopreservation outcomes in T cells (Hamann and Stathos, 2025a) and MSCs (Hamann and Stathos, 2025b). Here, it was evaluated as a cryoprotective agent for iPSCs by assessing post-thaw viability, health, expansion, and pluripotency. In all metrics tested, Optibumin 25 significantly outperformed clinical plasma-derived HSA.
RESULTS AND DISCUSSION
Greater Post-Thaw Viability and Improved Cell Health
To assess the effect of Optibumin 25 on iPSC cryopreservation, iPSCs grown in E8 FLEX media on vitronectin were harvested with Accutase and resuspended in various cryopreservation formulations: CryoStor CS10 alone, CS10 with 5% Optibumin 25, CS10 with 5% blood-derived HSA, CS5 alone, and CS5 with 5% Optibumin 25. All formulations included 10 µM Y-27632 ROCK inhibitor. Cells were frozen slowly at –80°C for 24 hours, transferred to liquid nitrogen for another 24 hours, then thawed and plated into two matching 96-well plates (one for expansion, one for flow cytometry) in E8 FLEX with ROCKi for 24 hours. Media was then changed to remove ROCKi, and viability dye plus annexin stain were added to one plate to assess cell viability (Figure 1A) and health (Figure 1B).
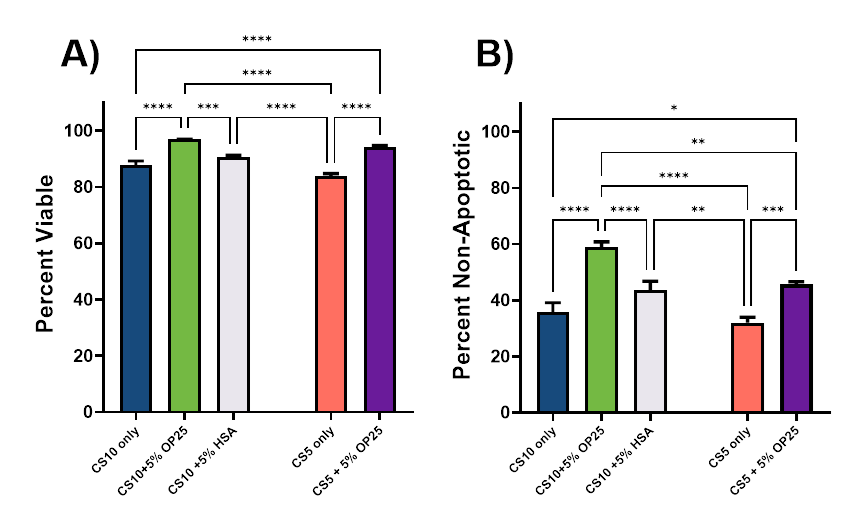
A) Viability – Characterization of 24-hour post-thaw viability in iPSCs frozen with CryoStor CS10 with or without 5%
Optibumin 25 or clinical blood-derived HSA or CryoStor CS5 with or without Optibumin 25 by high content imaging. (One Way ANOVA with Tukey post hoc test, p < 0.05). B) Health (percent non-apoptotic) in iPSCs frozen with CryoStor CS10 with or without 5% Optibumin 25 or blood-derived HSA or CryoStor CS5 with or without Optibumin 25 by high content imaging with a viability dye and Annexin V stain. (One Way ANOVA with Tukey post hoc test, p < 0.05).
As demonstrated in prior cryopreservation studies involving T cells and MSCs, the presence of 5% albumin improved viability in all conditions tested. While HSA provided a marginal and statistically insignificant benefit, 5% Optibumin 25 yielded a significant improvement over CS10 alone and CS5 alone, improving viabilities to greater than 90%. Surprisingly, the combination of CS5 and 5% Optibumin 25 significantly outperformed CS10 alone. These results indicate that Optibumin 25 not only improves performance but also allows DMSO reduction.
The apoptosis data followed a similar trend, though the benefit of adding Optibumin 25 to CryoStor 10 was more pronounced. This suggests viability improvements may be greater at later timepoints as stressed cells die.
The improved performance of Optibumin 25 compared to HSA may be attributed, in part, to its complete absence of stabilizers, such as sodium octanoate—a feature that differentiates it from serum-derived and yeast-derived albumins. These stabilizers can negatively impact sensitive cell types like iPSCs. Another contributing factor may be the higher free cys-34 content in Optibumin 25, which enhances its ability to reduce oxidative stress—one of the major challenges in iPSC cryopreservation (Lee et al., 2018).
Faster iPSC Recovery and Expansion with Optibumin 25
After the initial viability and cell health assessment, growth of the thawed iPSCs was monitored by high content imaging for an additional 24 hours. Cumulative fold expansion of viable cells was determined relative to the initial imaging scan taken immediately after addition of the viability, Annexin V, and nuclear stains (Figure 2).
As with the viability and cell health experiment, there is a significant improvement in fold expansion in groups cryopreserved with the addition of 5% Optibumin 25 compared to CS5 or CS10 alone and once again HSA fails to show a significant benefit. Here the benefit of CS5 and 5% Optibumin 25 is especially clear, resulting in a statistically significant 50% increase in cell expansion over 24 hours.
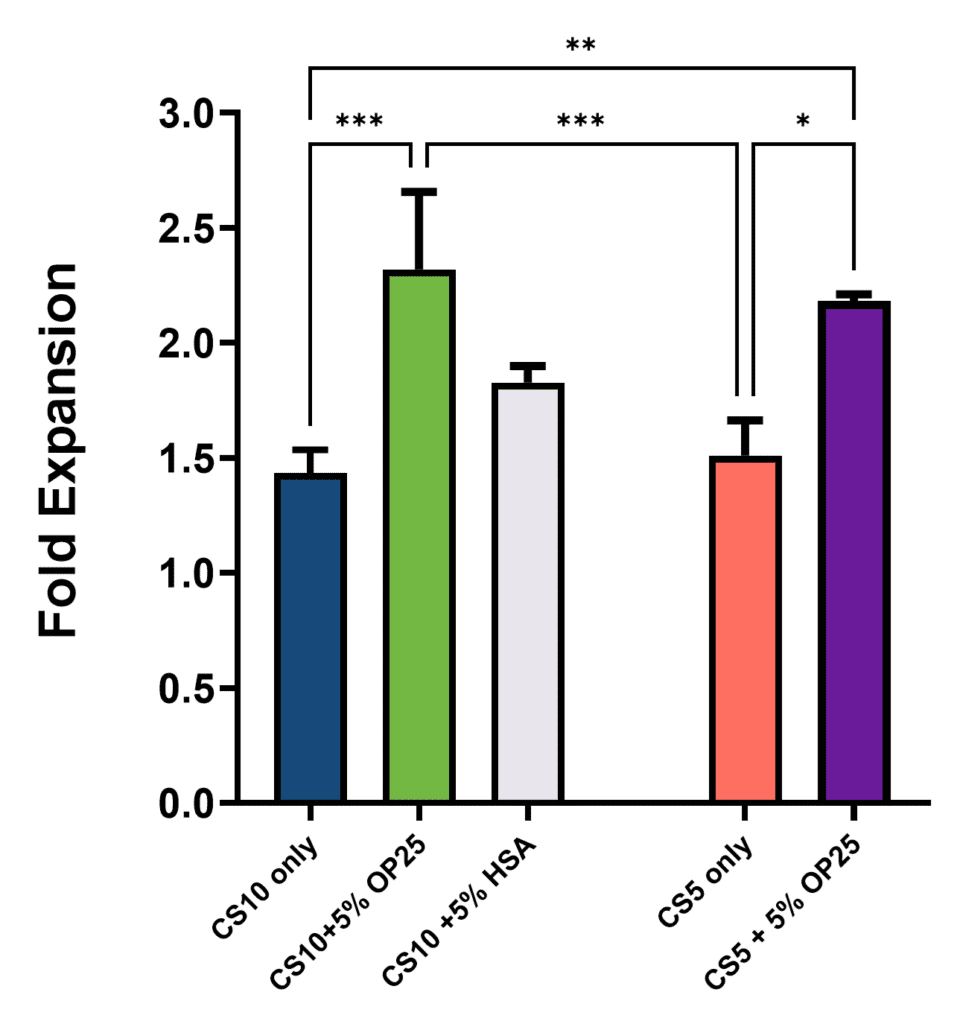
Characterization of 24 to 48 hour post thaw fold expansion by high content imaging for iPSCs frozen with CryoStor CS10 with or without 5% Optibumin 25 or blood-derived HSA or CryoStor CS5 with or without Optibumin 25. (One Way ANOVA with Tukey post hoc test, p < 0.05).
High Pluripotency Retention After Cryopreservation
To further characterize the effect of Optibumin 25 on iPSC post-thaw recovery, cells from the matching 96-well plate were analyzed by flow cytometry for the core pluripotency markers Oct4, Sox2, and Nanog—essential for iPSC maintenance (Rodda, 2005)—using a panel previously established for iPSC characterization. (Stathos and Hamann, 2025c) (See Figure 3).
Yet again, the benefit of Optibumin 25 was evident. Both conditions that included Optibumin 25 as well as the CS10 alone group maintained at least 90% pluripotency (defined here by Oct4, Sox2, and Nanog triple positivity) but the CS5 alone and CS10 with 5% HSA groups failed to reach this threshold and were significantly less pluripotent.
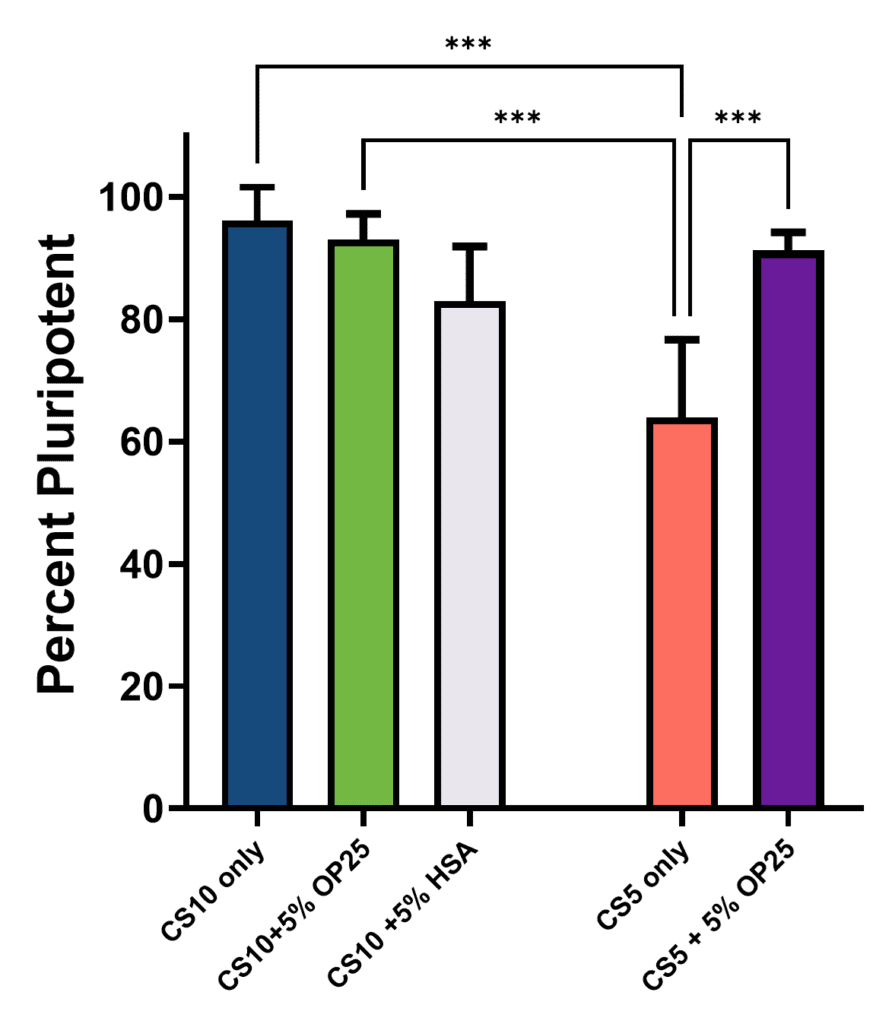
Flow cytometry analysis of Oct4+, Sox2+, and Nanog+ iPSCs following cryopreservation in CryoStor CS10 (±5% Optibumin 25 or blood-derived HSA) and CryoStor CS5 (±5% Optibumin 25). (One-way ANOVA with Tukey post hoc test, p < 0.05).
CONCLUSION
These findings support the integration of Optibumin 25 into iPSC cryopreservation protocols, particularly in workflows requiring high viability, pluripotency maintenance, and regulatory compliance for clinical manufacturing. Optibumin 25 significantly improved viability and cell health compared to those treated with blood-derived HSA. Additionally, supplementing CryoStor CS5 with 5% Optibumin 25 results in significant improvements in viability, cell health, and fold expansion while also reducing the DMSO concentration compared to the CryoStor CS10
benchmark. Taken together, Optibumin 25 offers significant improvement to iPSC cryopreservation by every metric tested, outperforms clinical HSA, and allows DMSO reduction while being chemically defined and animal origin free.
MATERIALS AND METHODS
Human iPSCs maintained in Essential 8™ FLEX media (ThermoFisher Scientific) were cultured in T flasks (Corning) coated with 0.5 µg/cm2 of N-terminal truncated Vitronectin (ThermoFisher Scientific) until approximately 75% confluent. The cells were then harvested by washing with PBS and treating with Accutase® (Innovative Cell Technologies, Inc) to singularize. The cells were counted using an NC-200 (ChemoMetec). For each condition, 1 × 10⁶ cells were aliquoted into microfuge tubes, pelleted by centrifugation and resuspended in one mL of the various cryopreservation formulations containing either Optibumin 25 or clinical grade blood-derived HSA with 10 µM Y-27632 ROCK inhibitor (StemCell Technologies). Cryopreservation formulations included base solutions of either CryoStor CS10 or CS5 (BioLife Solutions) used alone or
supplemented with either 5% Optibumin 25 or 5% blood-derived HSA. The cells were transferred to cryovials and frozen slowly in a Mr. Frosty™ (Thermo Fisher Scientific) passive freezing container for 24 hours at -80°C then transferred to liquid nitrogen for another 24 hours.
The next day, the cells stocks were thawed at 37°C and plated at 1 × 10⁵ cells per well in E8 FLEX with 10 µM ROCK inhibitor in two matching vitronectin coated 96 well plates. After 24 hours the media was changed to fresh E8 FLEX without ROCKi. A viability dye, an annexin (apoptotic) stain, and a live cell nuclear dye were added to one plate to characterize cell viability, health, and expansion via high content imaging. The viability and cell health data were taken immediately post staining and the fold expansion reflects growth over the next 24 hours.
After 24 hours of growth in E8 FLEX without ROCKi, the matching plate of cells was harvested for flow cytometry using Accutase. Cells were then stained with Zombie Aqua™ Fixable Viability Kit (Biolegend) and fixed and permeabilized using the FOXP3 / Transcription Factor Staining Buffer Set (eBioscience™, ThermoFisher Scientific). Cells were then stained with Oct4 Alexa Fluor® 647, Sox2 Alexa Fluor 488, and Nanog Alexa Fluor 594 antibodies (BioLegend) before being analyzed on a CytoFLEX LX® (Beckman Coulter) flow cytometer. Pluripotency was defined as the portion of live cells that were triple positive for Oct4, Sox2, and Nanog.
Featured Solution
Optibumin 25 – Recombinant Human Serum Albumin, 25% Solution – Animal-Origin-Free, GMP-Produced
Optibumin 25 is a chemically defined, recombinant albumin designed to enhance viability, reduce oxidative stress, and protect sensitive cells like iPSCs during cryopreservation. Its stabilizer-free formulation, high free cys-34 content, and animal-free origin make it an ideal supplement for serum-free cryopreservation protocols. Optibumin 25 supports safer, scalable biomanufacturing and regulatory compliance in cell and gene therapy, regenerative medicine, and vaccine development.
Frequently Asked Questions (FAQs)
Q: Why do iPSCs lose viability and pluripotency during cryopreservation, and how can this be prevented?
A: iPSCs are highly sensitive to cryopreservation stress, including osmotic and oxidative damage, and are negatively impacted by DMSO. Optibumin® 25 significantly improves post-thaw viability, expansion, and retention of pluripotency markers compared to plasma-derived HSA.
Q: Can DMSO concentrations be safely reduced in iPSC cryopreservation protocols?
A: Yes. Supplementing CryoStor® CS5 with 5% Optibumin® 25 enabled a reduction of DMSO from 10% to 5% while outperforming CryoStor CS10, supporting safer, lower-DMSO workflows.
Q: What advantages does recombinant albumin (Optibumin® 25) provide over plasma-derived HSA in cryopreservation?
A: Unlike blood-derived HSA, Optibumin® 25 is animal-origin-free, GMP-grade, chemically defined, and free of stabilizers that can harm sensitive cells. It reduces oxidative stress and improves viability, expansion, and pluripotency.
Q: How does Optibumin® 25 impact post-thaw recovery and expansion of iPSCs?
A: iPSCs cryopreserved with Optibumin® 25 showed faster recovery and a 50% increase in expansion post-thaw compared to HSA or CryoStor alone
Download the Full Application Note

The following content is gated. Please, subscribe to open access to it.
Footnotes
References
- Takahashi, K., & Yamanaka, S. (2006). Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell, 126(4), 663–676. https://doi.org/10.1016/j.cell.2006.07.024
- Sawamoto, N., Doi, D., Nakanishi, E., Sawamura, M., Kikuchi, T., Yamakado, H., Taruno, Y., Shima, A., Fushimi, Y., Okada, T., Morizane, A., Hiramatsu, S., Anazawa, T., Shindo, T., Ueno, K., Morita, S., Arakawa, Y., Nakamoto, Y., Miyamoto, S., Takahashi, R., & Takahashi, J. (2025). Phase I/II trial of iPS cell derived dopaminergic cells for Parkinson’s disease. Nature, 641(8064), 971–977. https://doi.org/10.1038/s41586-025-08700-0
- Reichman, T. W., Markmann, J. F., Odorico, J., Witkowski, P., Fung, J. J., Wijkström, M., Kandeel, F., de Koning, E. J. P., Peters, A. L., Mathieu, C., Kean, L. S., Bruinsma, B. G., Wang, C., Mascia, M., Sanna, B., Marigowda, G., Pagliuca, F., Melton, D., Ricordi, C., … Rickels, M. R. (2025, June 20). Stem cell–derived, fully differentiated islets for type 1 diabetes. New England Journal of Medicine. https://doi.org/10.1056/NEJMoa2506549
- Zhang, H., Wang, Q., Zhu, X., Xue, Y., Wang, J., & Wang, D. (2025). Reviving hearts, restoring lives: Long-term outcomes of allogeneic iPSC-cardiomyocytes transplantation for advanced heart failure patients. JACC: Basic to Translational Science, 10(3), 253–255. https://doi.org/10.1016/j.jacbts.2025.01.007
- Chang, C., Van Der Stegen, S., Mili, M., Clarke, R., Lai, Y.-S., Witty, A., Lindenbergh, P., Yang, B.-H., Husain, M., Shaked, H., Groff, B., Stokely, L., Abujarour, R., Lee, T., Chu, H.-Y., Pribadi, M., O’Rourke, J., Gutierrez, A., Riviere, I., … Valamehr, B. (2019). FT819: Translation of off-the-shelf TCR-less Trac-1XX CAR-T cells in support of first-of-kind phase I clinical trial. Blood, 134(Suppl 1), 4434. https://doi.org/10.1182/blood-2019-130584
- Dobruskin, M., Toner, G., & Kander, R. (2024). Optimizing cryopreservation strategies for scalable cell therapies: A comprehensive review with insights from iPSC-derived therapies. Biotechnology Progress, 40(5), e3504. https://doi.org/10.1002/btpr.3504
- Uhrig, M., Ezquer, F., & Ezquer, M. (2022). Improving cell recovery: Freezing and thawing optimization of induced pluripotent stem cells. Cells, 11(5), 799. https://doi.org/10.3390/cells11050799
- Sambo, D., Li, J., Brickler, T., & Chetty, S. (2019). Transient treatment of human pluripotent stem cells with DMSO to promote differentiation. Journal of Visualized Experiments, (149), e59833. https://doi.org/10.3791/59833
- Chen, G., Gulbranson, D. R., Hou, Z., Bolin, J. M., Ruotti, V., Probasco, M. D., Smuga-Otto, K., Howden, S. E., Diol, N. R., Propson, N. E., Wagner, R., Lee, G. O., Antosiewicz-Bourget, J., Teng, J. M. C., & Thomson, J. A. (2011). Chemically defined conditions for human iPSC derivation and culture. Nature Methods, 8(5), 424–429. https://doi.org/10.1038/nmeth.1593
- Hamann, A., & Stathos, M. (2025a). Enhanced cryopreservation of T cells using Optibumin®, recombinant human serum albumin (rHSA) [Application note]. InVitria. https://invitria.com/resources/enhanced-cryopreservation-of-t-cells-using-optibumin-rhsa/
- Hamann, A., & Stathos, M. (2025b). Low-DMSO cryopreservation of MSCs with Optibumin® 25 recombinant albumin [Application note]. InVitria. https://invitria.com/resources/low-dmso-cryopreservation-of-mscs-with-optibumin-25-recombinant-albumin/
- Lee, J., Cho, Y. S., Jung, H., & Choi, I. (2018). Pharmacological regulation of oxidative stress in stem cells. Oxidative Medicine and Cellular Longevity, 2018, 4081890. https://doi.org/10.1155/2018/4081890
- Rodda, D. J., Chew, J. L., Lim, L. H., Loh, Y. H., Wang, B., Ng, H. H., & Robson, P. (2005). Transcriptional regulation of Nanog by OCT4 and SOX2. Journal of Biological Chemistry, 280(26), 24731–24737. https://doi.org/10.1074/jbc.M502573200
- Stathos, M., & Hamann, A. (2025c). ITS-Animal-Free supplementation for iPSC expansion and pluripotency: A platform approach for serum-free cell expansion [Application note]. InVitria. https://invitria.com/resources/its-af-ipsc-expansion-pluripotency/