Optibumin® 25 – Recombinant human albumin
High-Purity Recombinant Albumin in Closed-System Bags for Advanced Biomanufacturing and Therapeutics
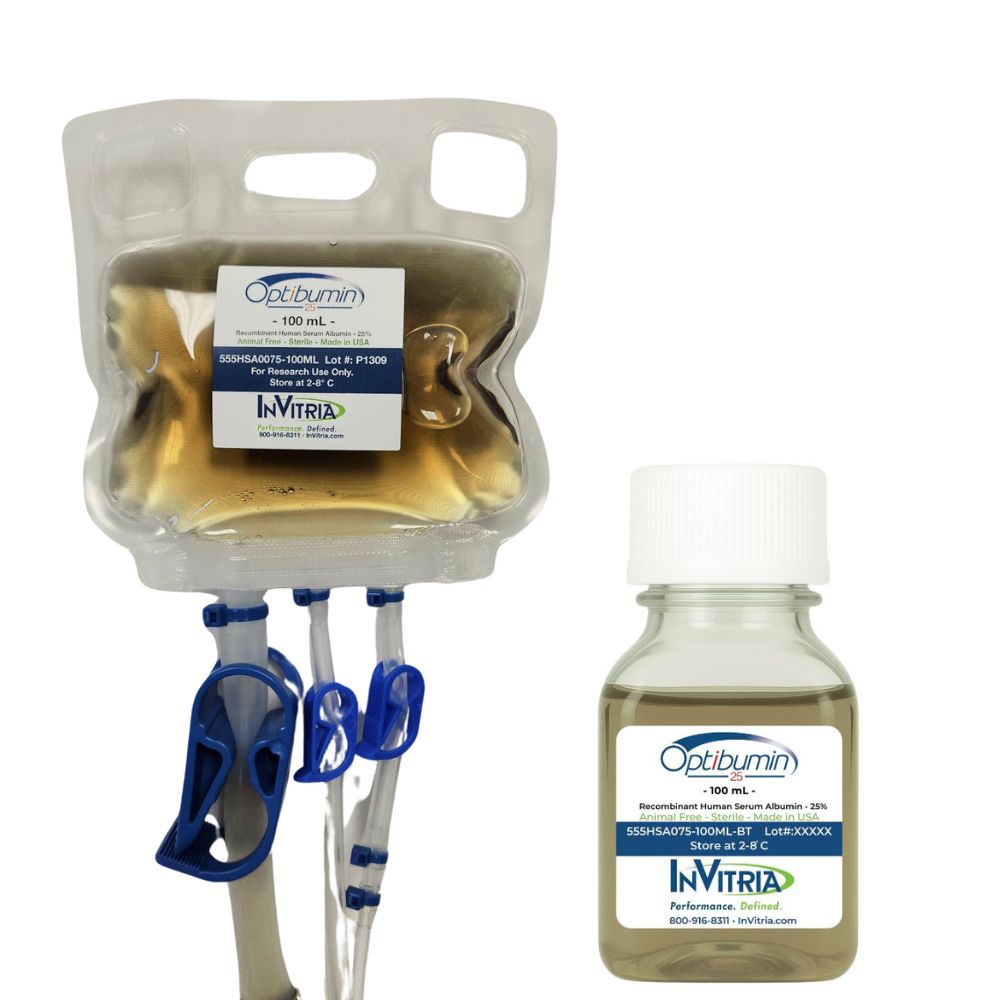

Product Name: Optibumin® 25 – Recombinant human albumin
Product Number: 555HSA0075
Product Form: 25% w/v Liquid
Name | Price | Qty | |
|---|---|---|---|
$785.00
|
Max:
Min: 1
Step: 1
|
||
$785.00
|
Max:
Min: 1
Step: 1
|
Product Description
Optibumin® 25 is a high-purity, recombinant human serum albumin (rHSA) at 25% concentration, designed to replace plasma-derived HSA in cryopreservation, cell therapy, and biomanufacturing applications. As the first recombinant, animal-origin-free 25% HSA solution available in a ready-to-use liquid format, Optibumin 25 eliminates the risks associated with blood-derived albumin, ensuring batch-to-batch consistency, pathogen safety, and regulatory compliance.
Free from stabilizers and preservatives, Optibumin 25 provides a chemically defined, scalable, and sustainable solution for industries requiring superior protein quality in critical workflows.<!--
Product Description
Optibumin is a lipid poor albumin that has been reduced to allow for maximal conjugation efficiency of different ligands for cell culture media optimization. Optibumin has exceptionally low endotoxin and high purity of mercaptoalbumin. Optibumin has been shown to be highly effective in certain cell culture applications such as human induced pluripotent stem cell expansion. This product is also suitable for use in formulation excipient and medical device applications.-->
Advantages
- Superior Purity and Performance
- Highest purity albumin available – free from stabilizers and preservatives.
- Low lipid content and high mercaptoalbumin (Cys34 free thiol) – essential for drug formulation, viral vector stabilization, and cryopreservation applications.
- Lowest product-related high molecular weight impurities – ensuring superior consistency and bioactivity.
- Animal-Origin-Free Assurance
- 100% chemically defined and animal-origin-free (AOF) – eliminates regulatory risks associated with blood-derived HSA.
- Manufactured using a non-animal, non-human host system, ensuring purity and reproducibility.
- Raw materials, manufacturing equipment, and packaging are fully animal-free, aligning with regulatory and ethical standards.
- Manufactured in the United States
- Produced in a tertiary-level, ISO 9001:2015 certified, animal-origin-free facility in the USA.
- Complete supply and manufacturing chain is owned and operated by InVitria, ensuring unmatched traceability and quality control.
- Consistent global supply, free from plasma donation limitations.
- Ready-to-Use Format
- Supplied as a sterile 25% liquid formulation in 100 mL closed-system friendly bags, eliminating the need for reconstitution.
- Seamlessly integrates into closed-system biomanufacturing workflows, reducing contamination risks and streamlining processing.
- Long-term stability and scalability for high-volume manufacturing needs.
Applications
- Cryopreservation & Cell Therapy
- Enables up to 40% DMSO reduction, improving cell viability and patient safety.
- Enhances T cell recovery and expansion, maintaining critical CAR-T memory phenotypes (Tscm, Tcm) post-thaw.
- Provides batch-to-batch consistency, ensuring reproducibility in cell therapy manufacturing.
- Gene Therapy & Vaccine Development
- Stabilizes viral vectors, supporting AAV, LV, and vaccine production.
- Prevents aggregation and loss of biological activity during long-term storage.
- Stem Cell Expansion & Regenerative Medicine
- Supports iPSC, MSC, and ESC culture, providing a chemically defined alternative to serum-derived albumin.
- Enhances cell growth and differentiation, eliminating concerns about plasma variability.
- Biopharmaceutical Formulations & Drug Conjugation
- High mercaptoalbumin (Cys34 free thiol) allows for superior small-molecule solubilization and drug conjugation.
- Ensures greater stability in recombinant protein formulations.
- In Vitro Fertilization (IVF) & Assisted Reproduction
- Ultra-low lipid content improves embryo culture conditions and developmental outcomes.
- Eliminates pathogen concerns associated with blood-derived albumin.
Technical Specifications
- Formulation: Sterile, ready-to-use 25% liquid solution
- Packaging: 100 mL closed-system friendly bags & 100 mL bottles
- Manufacturing: Produced in a state-of-the-art, ISO 9001:2015-certified facility in the USA
- Quality Assurance:
- Fully recombinant and animal-origin-free production system
- Stringent quality control processes ensure high batch-to-batch consistency
- Regulatory Compliance & Safety:
- cGMP-compliant manufacturing
- Plasma-free, pathogen-free, and chemically defined
- Aligned with regulatory expectations for closed-system biomanufacturing
Why Choose Optibumin 25?
Optibumin 25 sets a new standard for high-purity albumin solutions, offering unparalleled consistency, purity, and regulatory compliance compared to plasma-derived HSA. Whether for cryopreservation, cell therapy, vaccine production, or biopharmaceutical manufacturing, Optibumin 25 provides the safest, most scalable, and highest-performing alternative on the market.
See the Terms page for more information about the trademarks and patents covering this product.
Feedback
You must be logged in to post a review.
Reviews
There are no reviews yet.