Optibumin® 25 – Recombinant human albumin
High-Purity Recombinant Albumin in Closed-System Bags for Advanced Biomanufacturing and Therapeutics
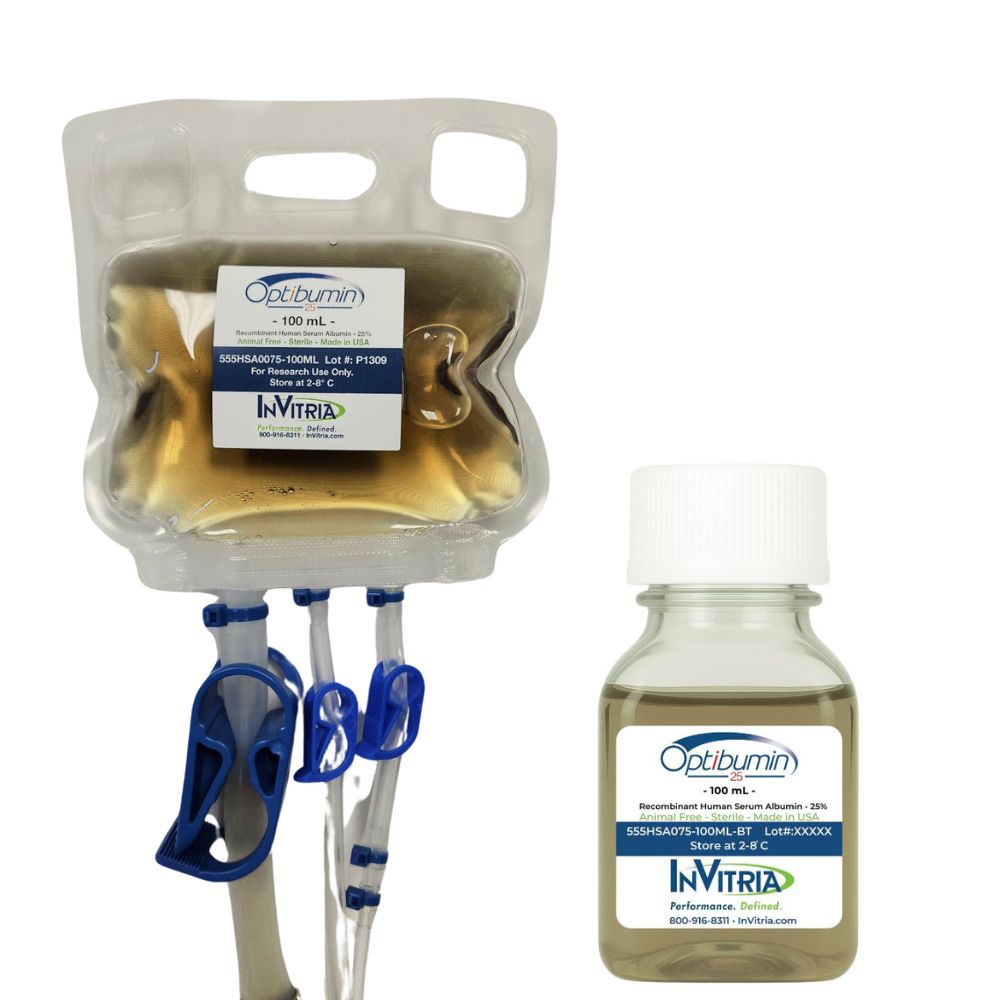

Product Name: Optibumin® 25 – Recombinant human albumin
Product Number: 777HSA106
Product Form: 25% w/v Liquid
Name | Price | Qty | |
|---|---|---|---|
$785.00
|
Max:
Min: 1
Step: 1
|
||
$785.00
|
Max:
Min: 1
Step: 1
|
Product Description
Optibumin® 25 is a high-purity recombinant human serum albumin (rHSA) supplied as a sterile 25% (w/v) liquid — the same concentration as plasma-derived human serum albumin — and is the only recombinant albumin available in closed-system bags for cell and gene therapy manufacturing. Produced using InVitria's animal-free ExpressTec expression platform at an ISO 9001:2015-certified, cGMP-compliant facility in Junction City, Kansas, Optibumin 25 is InVitria's recommended rHSA for most cell and gene therapy workflows.
Optibumin 25 delivers measurably superior performance compared to plasma-derived HSA: approximately 3x higher drug binding affinity, approximately 3x higher maleimide conjugation efficiency, and approximately 10x tighter lot-to-lot consistency across critical quality attributes. These performance advantages reflect a stabilizer-free, aggregate-depleted, native-state molecule with ≥99.0% purity — the highest purity available in the rHSA market — and approximately 99% reduced Cys34 thiol versus ~60% in plasma HSA.
With no added preservatives or stabilizers, no animal-derived components, and fully defined manufacturing from gene to final product, Optibumin 25 supports cryopreservation, cell therapy manufacturing, gene therapy and viral vector production, biopharmaceutical formulation, ADC development, drug solubilization, medical device applications, and diagnostic immunoassays. Optibumin 25 is available with full regulatory documentation — Certificate of Analysis, Safety Data Sheet, Certificate of Origin and Animal-Free / TSE-BSE Statement, ISO 9001 certificate, cGMP Statement, and regulatory-supporting packages — to accelerate IND, BLA, and supplier qualification workflows.
Advantages
Purity and Defined Composition
- ≥99.0% purity — the highest purity available in the rHSA market
- Endotoxin level ≤0.02 EU/mg
- Approximately 99% reduced Cys34 thiol — essential for controlled maleimide conjugation and drug binding (vs. ~60% reduced Cys34 in plasma-derived HSA)
- No added preservatives, no sodium octanoate, no N-acetyl tryptophan — unlike plasma-derived HSA, which requires stabilizers to survive pasteurization
- 100% animal-origin-free and blood-free — eliminates risks of adventitious viral, prion, and bacterial pathogens associated with plasma sourcing
Performance in Cell and Gene Therapy Manufacturing
- Enables up to 40% DMSO reduction in cryopreservation while maintaining CAR-T memory phenotypes (Tscm, Tcm) post-thaw (see T cell cryopreservation application note)
- Validated in primary T cell wash applications with >80% cell retention and <5% viability change post-wash (see T cell wash data)
- Preserves lentiviral titer during sterile filtration — a common loss point in viral vector manufacturing (see lentiviral filtration study)
- Supports iPSC cryopreservation with enhanced viability, expansion, and pluripotency preservation (see iPSC cryopreservation data)
- Available in closed-system bags with weldable tubing for seamless integration into sterile biomanufacturing workflows
Measurably Superior Performance vs. Plasma-Derived HSA
- Approximately 3x higher drug binding affinity at Sudlow site I (KD 2.58 µM vs. 7.45 µM; p < 0.01), demonstrated in head-to-head warfarin binding studies (see analytical characterization data)
- Approximately 3x higher maleimide conjugation efficiency, enabling defined-stoichiometry bioconjugation for ADC development (see conjugation study)
- Approximately 10x tighter lot-to-lot consistency across aggregate content, Cys34 redox state, and other critical quality attributes (standard deviation of aggregate fraction 0.20% vs. 3.57% for plasma HSA; F-test p < 0.0001)
- Up to 40% DMSO reduction demonstrated in T cell cryopreservation workflows while improving post-thaw viability (see low-DMSO T cell cryopreservation data)
Supply Chain Reliability and Manufacturing Control
- End-to-end vertically integrated US-based manufacturing at InVitria's ISO 9001:2015-certified, cGMP-compliant facility in Junction City, Kansas
- Consistent lot-to-lot performance enabled by recombinant expression, not donor-dependent sourcing
- Scalable to metric-ton production volumes without supply constraints from blood donation availability
- 3-year shelf life when stored at 2–8°C
Applications
Cryopreservation for Cell and Gene Therapy Optibumin 25 is validated in cryopreservation workflows for CAR-T, iPSC, and MSC therapies, enabling up to 40% DMSO reduction while improving post-thaw cell viability and preserving critical functional phenotypes. For CAR-T manufacturing, Optibumin 25 maintains Tscm and Tcm memory phenotypes post-thaw, supporting both patient safety during infusion (via reduced DMSO exposure) and preserved functional cell potency (see low-DMSO T cell cryopreservation data; see MSC cryopreservation data; see iPSC cryopreservation data). Its animal-origin-free, stabilizer-free, cGMP-manufactured profile supports raw material qualification for IND and BLA filings, and its lot-to-lot consistency ensures reproducibility across cryopreservation cycles in clinical and commercial manufacturing.
Cell Therapy Manufacturing and Cell Wash Optibumin 25 has been validated in primary T cell wash applications, achieving >80% cell retention across all conditions and post-wash viability changes of less than 5%, performing comparably to clinical-grade human serum albumin (see T cell wash application note). Its closed-system bag format integrates directly into sterile biomanufacturing workflows without transfer, reducing contamination risk and streamlining cell therapy process design. Compatible with CAR-T, TIL, stem cell, and other advanced therapy manufacturing platforms.
Gene Therapy and Viral Vector Manufacturing Optibumin 25 stabilizes viral vectors — including AAV, lentivirus (LV), and recombinant viral vectors — during upstream production, downstream processing, sterile filtration, and long-term storage. Published data demonstrate Optibumin 25 preserving lentiviral titer during sterile filtration, a common loss point in viral vector manufacturing (see lentiviral filtration study). Its defined protein environment prevents aggregation and preserves biological activity, supporting higher functional titers and extended shelf life in vaccine and gene therapy drug products.
Lentiviral Vector Manufacturing (CAR-T and Gene Therapy) Lentiviral vectors are notoriously fragile, with the lipid envelope vulnerable to shear, transmembrane pressure, and surface adsorption losses across every downstream unit operation — and final 0.22 µm sterile filtration is a particular pain point, with published losses exceeding 30% of infectious titer. In a recent InVitria application note (Stathos et al., 2026), pre-coating 0.22 µm PES filters with 1% (m/v) Optibumin 25 for just 5 minutes preserved approximately 75% of genomic titer (vs. 19% without Optibumin) and approximately 49% of infectious titer (vs. 17% without) — an approximately 3-fold increase in retained infectious titer. The protective mechanism combines shear protection, prevention of viral particle aggregation, and non-specific surface coating that reduces lentiviral adsorption to the filter membrane. Optibumin 25's excipient status — anchored by regulatory precedent in Merck's ERVEBO Ebola vaccine, which uses InVitria's recombinant albumin to stabilize VSV-G pseudotyped envelope virus — makes it suitable for both ex vivo CAR-T manufacturing and the emerging class of in vivo CAR-T therapies where lentiviral vector quality, consistency, and animal-origin-free regulatory profile are gating requirements. Applied at industrial scale, the 3-fold titer recovery improvement could translate into substantial cost-of-goods savings — published lentivirus production costs run approximately $100,000 per CAR-T dose in early clinical trials, meaning Optibumin 25 pre-treatment could reduce per-dose costs by an estimated $67,000.
Biopharmaceutical Formulation and Drug Binding Optibumin 25 stabilizes therapeutic proteins, monoclonal antibodies, and drug products by reducing aggregation, preventing surface adsorption, and maintaining protein activity in both liquid and lyophilized formulations. Head-to-head binding studies demonstrate approximately 3x higher drug binding affinity versus plasma-derived HSA (KD 2.58 µM vs. 7.45 µM at Sudlow site I, p < 0.01) (see analytical characterization data). Drugs complexed with Optibumin 25 experience a more significant reservoir effect and longer in vivo half-life than the same drugs with plasma HSA, enabling potentially lower or less frequent dosing — particularly meaningful for drugs with narrow therapeutic indices.
ADC Development and Bioconjugation Optibumin 25 is the preferred albumin scaffold for antibody-drug conjugates and other maleimide-based bioconjugation chemistry. With approximately 99% reduced Cys34 thiol (vs. ~60% in plasma HSA), Optibumin 25 delivers approximately 3x higher maleimide conjugation efficiency, enabling defined-stoichiometry conjugation and more predictable drug-to-albumin ratios (see covalent conjugation study). Its stabilizer-free composition (no sodium octanoate, no N-acetyl tryptophan) eliminates the allosteric interference with conjugation chemistry observed in plasma HSA.
Drug Solubilization and Hydrophobic Drug Delivery Albumin's high binding capacity for hydrophobic drugs — including two primary Sudlow sites, drug binding site III, and seven fatty acid binding sites — makes it an effective carrier for drugs that would otherwise require toxic organic solvents. Optibumin 25's stabilizer-free and aggregate-depleted structure maximizes accessible binding capacity, making it particularly well-suited for formulating poorly soluble small-molecule drugs, cytokines, and hydrophobic therapeutic payloads (see cytokine recovery and stability data).
Stem Cell Expansion and Regenerative Medicine Optibumin 25 supports iPSC, MSC, and other stem cell culture workflows, providing a chemically defined, animal-origin-free alternative to plasma-derived and serum-derived albumin. Its consistent lot-to-lot performance eliminates the variability that complicates stem cell research and manufacturing when using blood-sourced materials, and its defined profile supports regulatory qualification for cell therapy and regenerative medicine programs.
Medical Devices and Diagnostic Immunoassays Optibumin 25's chemically defined, animal-origin-free, stabilizer-free profile supports medical device applications including device coatings, blood-contacting surface treatments, and diagnostic device components. In diagnostic immunoassays, Optibumin 25 serves as a blocking agent that uniformly coats surfaces without interfering with specific binding events. In medical device coatings, albumin provides a biomimetic hemocompatible, antithrombogenic layer for blood-contacting surfaces. Both applications benefit from Optibumin 25's documented lot consistency.
In Vitro Fertilization and Assisted Reproduction Optibumin 25's ultra-low lipid content, chemically defined composition, and absence of stabilizers make it well-suited for embryo culture and assisted reproductive technology protocols. Its animal-origin-free profile eliminates the pathogen transmission concerns associated with serum-derived albumin traditionally used in IVF media, while delivering the consistent performance required for clinical reproductive medicine.
Optibumin 25 Technical Specifications
Key specifications for Optibumin 25 as supplied — including purity, endotoxin, formulation, storage, and manufacturing standards. Full Certificate of Analysis available upon request.| Specification | Details |
|---|---|
| Product Name | Optibumin® 25 |
| Brand | InVitria® |
| Catalog Number | 777HSA106 |
| Protein | Recombinant Human Serum Albumin (rHSA) |
| Class | Serum Protein / Albumin — Excipient Grade |
| Synonyms | rHSA, HSA, ALB, Albumin, Human Albumin, SA, rHA |
| Expression System | Recombinant, rice-based expression system |
| Molecular Weight | ~66.5 kDa |
| Purity | ≥ 99.0% |
| Endotoxin Level | ≤ 0.02 EU/mg |
| Formulation | Sterile 25% w/v (250 mg/mL) liquid |
| Concentration | 250 mg/mL (25% w/v) |
| Appearance | Slightly yellow/amber solution |
| Carrier Protein / Stabilizer | None (no additives or preservatives) |
| Sterility | Sterile (0.2 µm sterile filtered) |
| Packaging | 100 mL bag, 100 mL bottle |
| Storage Conditions | 2–8 °C |
| Shelf Life | 3 years |
| Animal-Origin-Free | Yes |
| Production Standard | ISO 9001:2015, cGMP-compliant facility |
| Country of Origin | USA (Junction City, KS) |
Why Select Optibumin 25 Over Plasma-Derived Albumin?
| Key Attribute | Optibumin 25 (rHSA) | Plasma-derived Albumin (pHSA) |
|---|---|---|
| FORMAT DIFFERENTIATORS | ||
| Available in closed-system bags | ✅ Yes (100 mL) | ❌ No |
| 25% (w/v) liquid — matches plasma HSA concentration | ✅ Yes | ✅ Yes |
| PERFORMANCE VS. PLASMA HSA | ||
| Drug binding affinity at Sudlow site I | ✅ ~3x higher (KD 2.58 µM vs. 7.45 µM, warfarin) | Baseline |
| Maleimide conjugation efficiency | ✅ ~3x higher | Baseline |
| Cys34 reduced thiol (preserved for bioconjugation) | ✅ ~99% reduced | ❌ Typically ~60% reduced (~40% cysteinylated) |
| Suitable for defined-stoichiometry bioconjugation | ✅ Yes (high Cys34 preservation) | ❌ Variable (high cysteinylation) |
| Aggregate burden (total aggregates) | ✅ <0.5% | ❌ Typically ~3–5%, varies by supplier |
| SAFETY PROFILE | ||
| Recombinant production | ✅ Yes | ❌ No (plasma-derived) |
| Animal-origin-free | ✅ Yes | ❌ Blood-derived |
| Blood-free | ✅ Yes | ❌ Blood-derived |
| Free of adventitious agent risk | ✅ Yes | ❌ Adventitious agent risk present |
| No donor variability | ✅ Yes | ❌ Donor-pool variability |
| TSE/BSE risk | ✅ None (no animal source) | ⚠️ Depends on donor screening |
| Suitable for defined-stoichiometry bioconjugation | ✅ Yes (high Cys34 preservation) | ❌ Variable (high cysteinylation) |
| FORMULATION ADDITIVES | ||
| Added preservatives | ✅ No | ❌ Typically contains preservatives, (sodium azide, sodium benzoate, potassium sorbate) |
| Added stabilizers | ✅ No | ❌ Typically contains stabilizers (sodium octanoate, N-acetyl tryptophan) |
| LOT-TO-LOT CONSISTENCY | ||
| Consistent lot-to-lot performance | ✅ Yes — controlled recombinant production | ❌ Variable — source-dependent |
| Performance traceable to a single manufacturing process | ✅ Yes — ExpressTec platform | ❌ Multi-donor plasma pool |
| SUPPLY CHAIN AND SCALABILITY | ||
| Scalable production (metric-ton capacity) | ✅ Yes | ❌ Limited by plasma donor supply |
| Supply independent of blood donation | ✅ Yes | ❌ Dependent on donor supply |
| Scale-up partnership with customer programs | ✅ Yes — InVitria scales with customer programs from clinical through commercial | ❌ Typically not provided |
| Vertically integrated supply chain | ✅ Yes — InVitria owns the complete supply chain | ❌ Multiple intermediaries |
| Manufactured in United States | ✅ Yes — Junction City, Kansas | Varies by supplier |
| Supports animal-free and sustainable sourcing mandates | ✅ Yes | ❌ No |
| REGULATORY AND DOCUMENTATION SUPPORT | ||
| ISO 9001:2015 certified manufacturing | ✅ Yes | Varies by supplier |
| cGMP-compliant QMS (21 CFR 210/211) | ✅ Yes | Varies by supplier |
| Certificate of Origin | ✅ Available | Varies by supplier |
| Animal-Free Statement | ✅ Available | ❌ Not applicable |
| Regulatory support package | ✅ Yes — full dossier-quality documentation for IND and BLA submissions | ❌ Limited or supplier-dependent |
| Technical support for regulatory filings | ✅ Yes — InVitria technical team supports customer regulatory submissions | ❌ Not typically provided |
| Full regulatory documentation on request | ✅ Yes | Varies by supplier |
Optibumin 25 Frequently Asked Questions (FAQs)
Product Overview
What Is Optibumin 25?
Optibumin 25 is a chemically defined, animal-origin-free, excipient-grade cGMP recombinant human serum albumin (rHSA) supplied as a sterile 25% (w/v) liquid — the only recombinant albumin available in closed-system bags and bottles suited for biomanufacturing. With ≥99.0% purity (the highest available in the rHSA market), no stabilizers, less than 0.5% total aggregates, and approximately 99% reduced Cys34 thiol, Optibumin 25 delivers the structural homogeneity and lot-to-lot consistency required for reproducible biomanufacturing and formulation.
What Is Optibumin 25 Made From?
Optibumin 25 is produced recombinantly using InVitria's ExpressTec expression platform, a non-mammalian, non-human expression host with no animal-derived, human-derived, or blood-derived materials. Manufacturing is fully animal-origin-free, from raw materials through production equipment to product-contact packaging.
Regulatory and Quality
Is Optibumin 25 USP Grade?
Optibumin 25 specifications are tested with consideration to the USP monograph for recombinant albumin (USP "rAlbumin Human"), with release specifications that exceed the purity expectations of the monograph. InVitria's analytical characterization enables more accurate quantitation of monomer purity and low-level aggregate content than standard compendial methods. Contact InVitria for full analytical and compendial documentation.
Is Optibumin 25 Excipient-Grade, i.e., Suitable for Final Formulation and Injectable Therapeutics?
Yes. Optibumin 25's specifications are well suited for use in final formulation of injectable biologics, vaccines, gene therapies, and cell therapies, supporting the regulatory requirements of IND/BLA-stage and commercial injectable drug product programs. Typical applications include viral vector stabilization (AAV, LVV), live-attenuated vaccine formulation, cryopreservation formulations, and biopharmaceutical drug product formulation where defined, blood-free albumin with low endotoxin and high purity is required for injectable use.
Is Optibumin 25 cGMP-Manufactured?
Yes. InVitria manufactures Optibumin 25 under a cGMP-compliant Quality Management System (QMS) with respect to 21 CFR Part 210/211 at its ISO 9001:2015-certified facility in Junction City, Kansas, USA.
What Quality Documentation Is Available for Optibumin 25?
Optibumin 25 is supported by a Certificate of Analysis, Safety Data Sheet, Guidelines for Use, Certificate of Origin and Animal-Free / TSE-BSE Statement, ISO 9001 certificate, and cGMP Statement. Additional Quality and Regulatory documentation is available on request — including materials for IND-supporting packages and supplier qualification.
Does Optibumin 25 Contain Stabilizers?
No. Optibumin 25 is supplied without sodium octanoate (caprylate), N-acetyl tryptophan, or other stabilizers. This is a fundamental difference from plasma-derived HSA, which requires 16 mM each of sodium octanoate and N-acetyl tryptophan to survive pasteurization for pathogen inactivation.
Selection and Comparison
What Is the Difference Between Optibumin 25 and Optibumin 20?
Optibumin 25 and Optibumin 20 are both liquid recombinant human serum albumin (rHSA) products manufactured by InVitria using the same ExpressTec expression platform, sharing the same ≥99.0% purity specifications and animal-origin-free profile. Optibumin 20 is a 20% (w/v) liquid supplied in bottles, designed for general upstream process use in cell and gene therapy workflows, cell culture supplementation, hydrophobic molecule solubilization, and select clinical applications. Optibumin 25 is a 25% (w/v) liquid — the same concentration as plasma-derived human serum albumin — supplied in both bottles and closed-system bags.
For cell and gene therapy manufacturing workflows — particularly closed-system workflows, cryopreservation, and applications where Optibumin is used as a direct alternative to 25% plasma-derived HSA — Optibumin 25 is the recommended product. For general upstream process applications where a 20% liquid albumin in bottles is sufficient, Optibumin 20 is appropriate. Contact InVitria to discuss the best option for your specific application.
What Is the Difference Between Optibumin 25 and Exbumin?
Optibumin 25 and Exbumin are both excipient-grade rHSA products manufactured by InVitria using the same ExpressTec expression platform. Both are rice-derived, chemically defined, animal-origin-free, and suitable for use as excipients in final drug formulation. The primary difference is format.
Exbumin is supplied as a lyophilized powder (≥98.0% purity) and carries documented regulatory precedent as a formulation component in Merck's ERVEBO® Ebola vaccine, with acceptance as an excipient by the FDA, EMA, and PMDA. Optibumin 25 is supplied as a sterile 25% (w/v) liquid (≥99.0% purity, the highest available in the rHSA market) in both bottles and closed-system bags, designed for cell therapy, gene therapy, cryopreservation, cell wash, and applications using rHSA as a direct alternative to 25% plasma-derived HSA.
Choose Exbumin when a lyophilized powder is required for formulation or stability. Choose Optibumin 25 when a liquid rHSA — particularly in closed-system packaging — best fits the manufacturing workflow. Contact InVitria to discuss the best option for your specific application.
What Is the Difference Between Optibumin 25 and Cellastim S?
Optibumin 25 is a liquid rHSA designed for cell therapy, gene therapy, cryopreservation, and a broad range of biomanufacturing applications. Cellastim S is the cell culture-grade rHSA designed for serum-free media supplementation, with specifications appropriate for that application. Reach out to InVitria's Product Applications Team to receive suggestions on the best fit.
How Is Optibumin 25 Different From Yeast-Derived Recombinant Albumins?
Yeast-derived recombinant albumins addressed the pathogen risk of plasma HSA but introduced new issues: immunogenic yeast host cell proteins requiring extensive purification, and surfactants like Polysorbate 80 added to prevent processing aggregation. Optibumin 25's non-mammalian, non-yeast ExpressTec expression system produces a native-state molecule without these constraints.
When Should I Use Optibumin 25 Instead of Plasma HSA?
Choose Optibumin 25 over plasma HSA when your application depends on predictable drug binding affinity, high-efficiency maleimide conjugation, low aggregate burden, tight lot-to-lot consistency, animal-origin-free raw material qualification, or freedom from stabilizers that interfere with binding or analytical methods.
Application and Use
Can Optibumin 25 Be Used in Cell Therapy Manufacturing?
Yes. Optibumin 25 has been validated in primary T cell wash applications, performing comparably to clinical-grade human serum albumin in cell retention (>80% across all conditions) and viability (changes less than 5% post-wash). Its animal-origin-free status and consistent lot-to-lot quality support raw material qualification for IND and BLA filings.
Can Optibumin 25 Be Used in Cryopreservation for Cell and Gene Therapy?
Yes. Optibumin 25 is used in cryopreservation formulations for advanced therapies including CAR-T, iPSC, and MSC workflows, and has been shown to enable up to 40% DMSO reduction while improving post-thaw cell viability. It supports T cell recovery and expansion while maintaining critical CAR-T memory phenotypes (Tscm, Tcm) post-thaw. For cell therapy developers, reduced DMSO exposure translates directly into improved patient safety during infusion and better preservation of functional cell phenotype, and batch-to-batch consistency ensures reproducibility across cryopreservation cycles in clinical and commercial manufacturing.
Can Optibumin 25 Be Used to Stabilize Viral Vectors?
Yes. Optibumin 25 stabilizes viral vectors including AAV and lentivirus (LVV) in gene therapy and vaccine production workflows. Its defined protein environment prevents aggregation and preserves biological activity during production, sterile filtration, and long-term storage, supporting higher functional titers and extended shelf life for viral vector drug products. Published InVitria data show Optibumin 25 preserving lentiviral titer during sterile filtration, a common loss point in viral vector manufacturing. Optibumin 25 is suitable for evaluation in both upstream viral vector stabilization during production and downstream final formulation of injectable gene therapy drug products.
Can Optibumin 25 Be Used for Albumin-Drug Conjugates and ADCs?
Yes. Optibumin 25 is particularly well-suited for ADC applications. Its approximately 99% reduced Cys34, approximately 3x higher maleimide conjugation efficiency versus plasma HSA, and absence of stabilizers make it the preferred scaffold for defined stoichiometry conjugation chemistry.
Can Optibumin 25 Be Used for Drug Solubilization?
Yes. Albumin's high binding capacity for hydrophobic drugs — including two main Sudlow sites, drug binding site III, and seven fatty acid binding sites — makes it an effective carrier for drugs that would otherwise require toxic organic solvents. Optibumin 25's stabilizer-free and aggregate-depleted structure maximizes the accessible binding capacity.
Can Optibumin 25 Be Used in Medical Device Applications and Diagnostic Immunoassays?
Yes. Optibumin 25's chemically defined, animal-origin-free, stabilizer-free profile makes it well-suited for medical device applications including device coatings, blood-contacting surface treatments, and diagnostic device components. Its structural homogeneity and lot-to-lot consistency support reproducible device performance where albumin lot variability would otherwise compromise product design.
Can Optibumin 25 Be Used in Diagnostic Immunoassays?
Yes. Optibumin 25's chemically defined, animal-origin-free, stabilizer-free profile makes it well-suited for medical device applications including device coatings, blood-contacting surface treatments, and diagnostic device components. In diagnostic immunoassays, it serves as a blocking agent that uniformly coats surfaces without interfering with specific binding events. In medical device coatings, it provides a biomimetic hemocompatible, antithrombogenic layer for blood-contacting surfaces. Both applications benefit from Optibumin 25's documented lot consistency.
Analytical Properties
Does Optibumin 25 Bind Drugs Better Than Plasma HSA, and Why?
Yes. In head-to-head binding studies with warfarin — a model drug binding Sudlow site I — Optibumin 25 demonstrated a KD of 2.58 µM versus 7.45 µM for plasma HSA, representing approximately 3x higher binding affinity (p < 0.01, Fisher's F-test). Optibumin 25's measured affinity aligned with literature expectations, while plasma HSA bound significantly worse than predicted.
Three factors contribute to this difference: Optibumin 25 contains no sodium octanoate or N-acetyl tryptophan — the plasma HSA stabilizers that bind Sudlow site II and allosterically interfere with drug binding at Sudlow site I; it has 10x fewer aggregates than plasma HSA, preserving accessible surface area for drug interaction; and it has approximately 99% reduced Cys34 versus approximately 60% in plasma HSA, where cysteinylation is known to reduce warfarin binding.
The practical implication: drugs complexed with Optibumin 25 experience a more significant reservoir effect and longer in vivo half-life than the same drugs complexed with plasma HSA, enabling potentially lower or less frequent dosing — particularly meaningful for drugs with narrow therapeutic indices.
How Consistent Is Drug Binding Across Optibumin 25 Lots?
Optibumin 25 demonstrates approximately 10-fold tighter lot-to-lot consistency than plasma HSA across aggregate content, Cys34 redox state, and other critical quality attributes. Standard deviation of aggregate fraction is 0.20% for Optibumin 25 versus 3.57% for plasma HSA (F-test for variance, p < 0.0001).
What Is the Cys34 Redox State of Optibumin 25, and How Does It Affect Maleimide Conjugation?
Optibumin 25 has approximately 99% reduced (unmodified) Cys34 across 10 production lots, compared to approximately 60% reduced Cys34 in plasma HSA. Cys34 is albumin's single free cysteine and the reactive handle used for maleimide-based bioconjugation in ADC development. A preserved Cys34 thiol is essential for controlled, stoichiometric maleimide conjugation — cysteinylated Cys34 cannot participate, so plasma HSA's 40% cysteinylation translates directly into lower conjugation efficiency and less predictable drug-to-albumin ratios.
Combined with the absence of stabilizers that can interfere with conjugation chemistry, this preserved thiol enables Optibumin 25 to achieve approximately 3x higher maleimide conjugation efficiency compared to plasma-derived HSA.
Packaging and Ordering
What Packaging Options Are Available for Optibumin 25?
Optibumin 25 is available in 100 mL closed-system-friendly bags with weldable tubing, as well as 100 mL bottles. The closed-system bag format is engineered to integrate directly into sterile biomanufacturing workflows without requiring transfer. Contact InVitria for bulk and custom packaging options.
Storage and Handling
How Should Optibumin 25 Be Stored?
Store Optibumin 25 at 2–8°C per the Guidelines for Use and product Certificate of Analysis. Handle aseptically when removing aliquots from the primary container and minimize temperature excursions to preserve product performance. Shelf life is 3 years from date of manufacture. Contact InVitria technical support for application-specific handling guidance.
Peer-Reviewed Research Citing Optibumin
Optibumin has been cited in peer-reviewed publications spanning vaccine development, gene therapy, cell therapy, and clinical diagnostics. Here are the most-cited papers referencing Optibumin usage, including distribution catalog numbers| Paper Title | Journal/Publisher | Year |
|---|---|---|
| Transgene Biocontainment Strategies for Molecular Farming | Frontiers in Plant Science | 2020 |
| Exploring recent progress of molecular farming for therapeutic and recombinant molecules in plant systems | Heliyon | 2024 |
| Biotechnologically Engineered Plants | Biology | 2023 |
| Effects of recombinant human albumin as a culture media supplement on mouse embryo development | Fertility and Sterility | 2021 |